News
Baker’s yeast the secret behind 15-minute COVID test
A new type of COVID-19 screening being developed by researchers in Queensland could provide results faster than a pregnancy test.
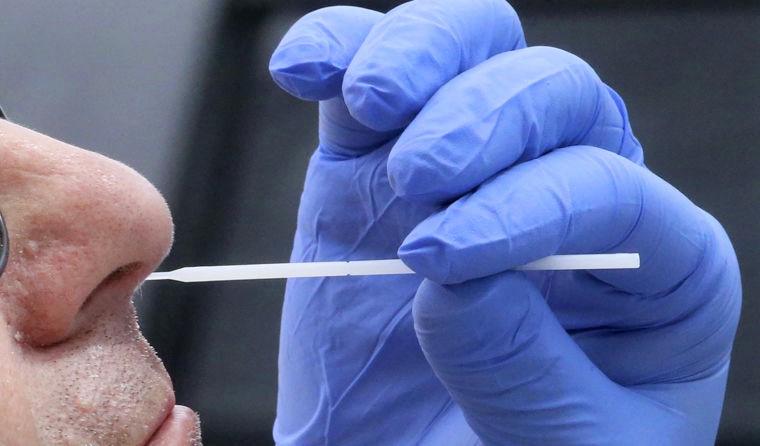 The new test could potentially produce results from nasal swabs within 15 minutes. (Image: AAP)
The new test could potentially produce results from nasal swabs within 15 minutes. (Image: AAP)
The test, called XavTrap, has attracted millions in funding at home and abroad, with the ongoing research and development being conducted by biotechnology firm Xing Technologies.
The Queensland Government recently contributed $1.5 million to its development, while the US Food and Drug Administration (FDA) has also backed the project with $1.4 million (US$1 million).
While the majority of point-of-care coronavirus tests to date have relied on serology, researcher Dr Yadveer Grewal explains that XavTrap combines the use of nasal swabs with a cheap and simple baker’s yeast, which is coated with hook-like particles to trap the virus, similar to velcro.
That yeast is then combined with strip technology used in pregnancy tests to rapidly determine whether a person has the virus.
‘That way someone could have a nasal swab taken, apply it to a strip and within 5–15 minutes, depending on how infectious they are, get a result then and there,’ he told Nine’s Today program.
There is currently a risk that high-quality nasal swab lab tests can produce false-negatives.
But Dr Yadveer said the new test could still be effective, even though it is less sensitive, and that the FDA believes the sensitivity and price of tests can be lowered if people using them get tested more frequently.
To expand on his point, Dr Yadveer used the example of doctors and nurses undergoing Xing’s test every time they started a hospital shift and getting a fast result.
‘Testing yourself every couple of days is about equivalent to having a once-off high-quality specific test from a lab,’ he said.
Another difference between XavTrap and other tests is that it discriminates between live and dead viruses, so it is said to be less likely to result in false-positives.
Associate Professor Mark Morgan, Chair of the RACGP Expert Committee – Quality Care (REC–QC), previously told newsGP point-of-care coronavirus testing would be useful in a general practice setting.
‘If GPs were able to identify patients who have protective immunity … this would be game-changing,’ he said.
‘However, any new piece of testing technology needs to have clear descriptions of the ways it is intended to be used. We don’t want to have a repeat of the current misuse [of] point-of-care serology testing.’
Xing Technologies wants to have its new COVID-19 test on the market by the end of 2020, and hopes to eventually use XavTrap to test for other infectious viruses like dengue fever, along with non-communicable diseases such as cancer.
‘Ideally, then, we are able to manufacture immediately by the end of the year and distribute it,’ Dr Yadveer said.
‘Because our technology is easily programmable we have other lead candidates we are exploring, and they can come online early next year.’
With AAP
Log in below to join the conversation.
COVID-19 Queensland rapid test
newsGP weekly poll
Health practitioners found guilty of sexual misconduct will soon have the finding permanently recorded on their public register record. Do you support this change?