News
Possible RSV vaccine is ‘very exciting’
Given to pregnant women, the RSV vaccine is shown to prevent severe disease for up to the first six months of life.
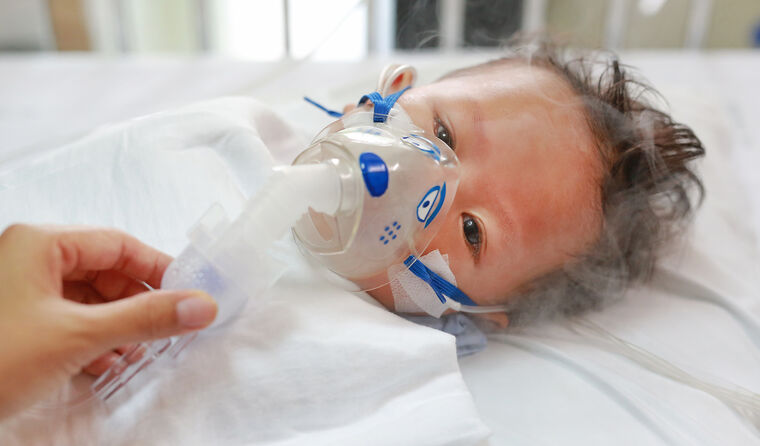 The burden of RSV falls most heavily on very young children, particularly premature babies and those in the first few months of life.
The burden of RSV falls most heavily on very young children, particularly premature babies and those in the first few months of life.
Respiratory syncytial virus (RSV) is responsible for more than 100,000 deaths and 3.6 million hospitalisations in children worldwide every year.
Development of a vaccine has been in the works since the 1960s, and new research published in the New England Journal of Medicine suggests those efforts may finally soon bear fruit.
According to the report, researchers have developed an RSV vaccine that when given to pregnant women is shown to be 81.8% effective in preventing severe disease within 90 days of birth and 69.4% effective in preventing severe disease within 180 days of birth.
Dr James Best, Chair of RACGP Specific Interests Child and Young Person’s Health, told newsGP the ‘interesting and exciting’ study shows promise, with the very young – particularly premature babies and those in the first few months of life – likely to benefit.
‘In general practice, if I see a baby in the first few months of life with a viral illness causing respiratory distress, I hope that it isn’t RSV … the younger you are, the greater the risk,’ he said.
‘Complications from RSV in these populations are not uncommon, and the hospitalisation rate in infants 0–2 months of age was estimated in 2019 at 2778 per 100,000 population.
‘This is why emergency departments will often screen for RSV via PCR testing in these situations, as well as for monitoring population trends.’
RSV infects the airways and lungs and can lead to life-threatening complications such as bronchiolitis and pneumonia, making it one of the leading causes of hospitalisation for babies globally.
The double-blind, phase three trial was conducted in 18 countries, including Australia, and comprised 7358 pregnant women.
Half were given a single intramuscular injection of 120 mcg of a bivalent RSV prefusion F protein-based (RSVpreF) vaccine and half were given a placebo between 24 and 36 weeks of pregnancy.
Within 90 days of birth, six infants in the vaccine group developed severe lower respiratory tract illness compared to 33 in the placebo group, equating to 81.8% efficacy. Within 180 days of birth there were 19 and 62 cases respectively, equating to 69.4% efficacy.
Dr Kerry Hancock, Chair of RACGP Specific Interests Respiratory Medicine, told newsGP there are other benefits to an RSV vaccine in addition to preventing severe illness.
‘This is very exciting – we are so close now to having a vaccine that can reduce the burden of RSV illness in infants where the burden of disease is most pronounced,’ she said.
‘[It will mean] less infants with bronchiolitis, less ear infections, less bacterial pneumonias that are associated with RSV infection in young children, less antibiotics, and other benefits [like] preventing longer term complications such as chronic lung infections and asthma.’
Dr Hancock says 34 years ago her own 10-week-old son had severe bronchiolitis requiring admission to hospital.
‘The prospect of a vaccine that could prevent that gives me joy that another mother does not have to go through that drama, stress and worry,’ she said.
While RSV was discovered in 1956, it only became a notifiable condition in Australia in 2021. And in recent years, its patterns have been affected by COVID and lockdowns, according to Dr Best.
‘RSV has had an unusual rollercoaster ride through and after the pandemic,’ he said.
‘For a while it virtually disappeared, but then had a surge recorded in summer months in some parts of Australia, when it normally has a winter to early spring peak.’
Dr Hancock says non-pharmaceutical interventions like hand washing, social distancing, isolation, mask wearing and working from home also had an impact on RSV trends.
‘[All of these] impacted on the circulation of most respiratory viruses including RSV globally, particularly in Australia with the absence of the normal winter epidemic in 2020,’ she said.
‘Sentinel hospital records showed a marked decline in RSV related hospitalisation in the winter of 2020.
‘Then RSV activity rebounded in late 2020 [spring] and early 2021 [summer] with unprecedented widespread RSV outbreaks occurring in NSW, ACT and WA coinciding with the easing of COVID restrictions in those geographical regions.
‘Hospital admission levels in those unseasonal times were reported to be equivalent or exceeding the normal winter seasonal RSV activity in any of the previous three years.’
As we move further away from the peak of the COVID pandemic, Dr Best thinks traditional RSV patterns will once again come to the fore.
And while an RSV vaccine could minimise future outbreaks, the trial is currently only in phase three, which means it is not yet approved for consumers.
Another identified limitation of the study was that women with high-risk pregnancies were excluded, meaning those with multiple pregnancy, risk of pre-term birth or a previous infant with a significant congenital anomaly were omitted, even though those children are at more risk of severe RSV illness.
Nonetheless, Dr Best is hopeful that parents will soon have a new way of protecting their young children from the potentially serious consequences that RSV can pose.
‘Approving and recommending a vaccine is a complicated and highly regulated process, and so it should be,’ Dr Best said.
‘But if we are getting closer to a vaccine available to Australian pregnant women to help protect their infants-to-be from what can be a devastating disease, well that is very good news indeed.’
Log in below to join the conversation.
respiratory syncytial virus RSV vaccine
newsGP weekly poll
As a GP, do you use any resources or visit a healthcare professional to support your own mental health and wellbeing?