News
Shortage of diabetes drug flagged until 2023
While semaglutide supply will be intermittent, its current availability should be ‘enough for approved use’, the manufacturer has advised.
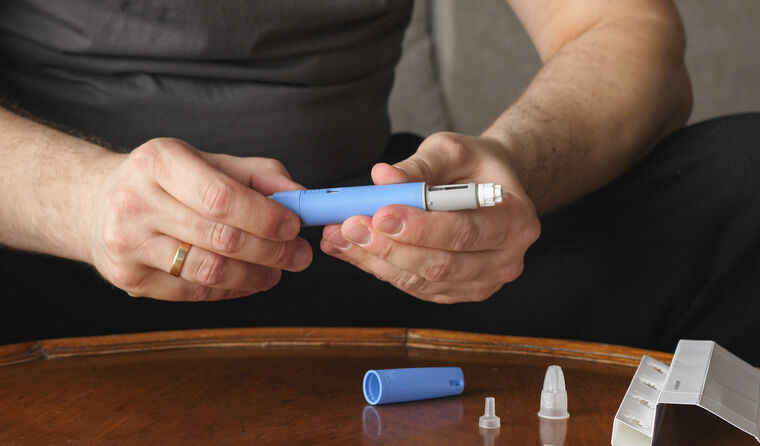 A patient prepares an injection of semaglutide, which is likely to be in short supply for the rest of the year.
A patient prepares an injection of semaglutide, which is likely to be in short supply for the rest of the year.
Shortages of the diabetes drug semaglutide (Ozempic) are likely to extend at least until the end of the year, the Therapeutic Goods Administration (TGA) has warned.
In a joint statement issued earlier this month, the TGA said there will likely be ‘intermittent availability’ of the treatment due to an unexpected increase in consumer demand.
As with previous shortages the demand ‘is due to extensive prescribing for obesity management, for which Ozempic is not indicated,’ the statement said.
‘The shortage is significantly affecting people using Ozempic for its approved use for type 2 diabetes.’
The treatment’s sponsor Novo Nordisk Pharmaceuticals, the RACGP, and several other organisations including the Pharmaceutical Society of Australia and Diabetes Australia co-issued the statement with the TGA.
A shortage of another type 2 diabetes treatment, dulaglutide 1.5 mg (sold as Trulicity), which has been driven by the supply issues affecting semaglutide, looks likely to ease at the end of this month.
Dr Gary Deed, Chair of RACGP Specific Interests Diabetes, previously told newsGP that the treatments should be restricted to those with type 2 diabetes.
‘There are [other] TGA-approved medications for management of obesity,’ he said.
‘These always should be used in a framework of lifestyle and diet changes, and liraglutide is a suitable alternative GLP-1RA [glucagon-like peptide-1 receptor agonist] given daily for weight and obesity management.
‘It is important to allow access of people with type 2 diabetes to usual medication – and this should be a medical priority.’
Health professionals continue to be urged by the TGA to limit the prescribing and dispensing of semaglutide for approved use only to people with type 2 diabetes.
This is defined by the TGA as the treatment of adults with insufficiently controlled type 2 diabetes mellitus as an adjunct to diet and exercise:
- as monotherapy when metformin is not tolerated or contraindicated
- in addition to other medicinal products for the treatment of type 2 diabetes.
The sponsoring company said there should be sufficient supplies if it is prescribed for diabetes patients only, and that it is taking steps to reduce the impact of off-label prescribing.
It is also encouraging patients to leave prescriptions with their regular pharmacy if possible.
‘This will allow the pharmacist to order it for you and help you to get the medicines you need,’
Novo Nordisk states on its website.
‘We are also strongly encouraging pharmacists to limit dispensing to only one month of medication at a time.’
The TGA has warned consumers that if they have a prescription for semaglutide but do not have type 2 diabetes, their ‘pharmacist may not be able to fill your prescription because of the current shortage’.
The
Australian Journal of Pharmacy recently reported that pharmacists are seeing little reduction in off-label prescribing despite ongoing requests from the TGA to limit non-approved use.
Demand for the treatments, which act as an appetite suppressant, has increased significantly this year following widespread promotion on social media for weight loss.
Log in below to join the conversation.
diabetes dulaglutide Ozempic semaglutide Trulicity
newsGP weekly poll
How often do patients ask you about weight-loss medications such as semaglutide or tirzepatide?