Feature
Safety concerns over popular arthritis drug
The TGA issued an alert after a study found high doses of tofacitinib could lead to increased risk of blood clots in the lungs and potentially death.
 The Therapeutic Goods Administration has advised doctors should monitor patients for signs and symptoms of pulmonary embolism.
The Therapeutic Goods Administration has advised doctors should monitor patients for signs and symptoms of pulmonary embolism.
Pharmaceutical manufacturer Pfizer is in the process of alerting doctors of the risk, after a clinical trial revealed the potential dangers for patients taking a 10 mg twice-daily dose of tofacitinib (sold as xeljanz) for the treatment of rheumatoid arthritis.
Tofacitinib generated revenue of $2.5 billion last year, but a post-marketing study designed to assess whether it increased the risk of cardiovascular events in patients older than 50 resulted in a ‘notification’ from the rheumatology Data Safety Monitoring Board (DSMB) about the 10 mg dosing arm in the trial.
The Therapeutic Goods Administration (TGA) issued a safety alert yesterday in which it advised it is ‘closely monitoring’ the situation and working with Pfizer Australia to investigate the findings.
It also said doctors should monitor patients for signs and symptoms of pulmonary embolism, and follow recommendations outlined in the product information in relation to the specific conditions for which tofacitinib is used.
‘A 10 mg twice-daily dose is not approved for treatment of rheumatoid arthritis in Australia. However, that dose is approved in Australia for the initial and short-term treatment of adult patients with moderately to severely active ulcerative colitis,’ the TGA alert stated.
‘It is not recommended as a first-line treatment, meaning it is approved for use in ulcerative colitis patients for whom other treatments are not appropriate, have not worked, or have lost effectiveness.’
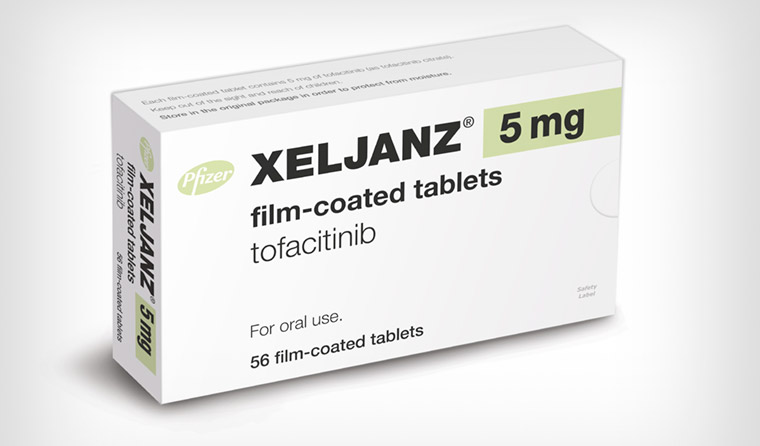
Tofacitinib (sold as xeljanz) generated around $2.5 billion in sales last year.
Pfizer first revealed the dangers in February, when it announced it had ‘taken steps to transition’ patients on tofacitinib at 10 mg twice daily to a reduced 5 mg twice-daily dose, after the DSMB found the higher dosage group had a ‘statistically and clinically important difference’ in the occurrence of pulmonary embolism.
The post-marketing study was designed to assess the risk of cardiovascular events, such as heart attack, coronary artery disease, heart failure and abnormal heart rhythms, in patients aged at least 50 years who had at least one other cardiovascular risk factor. All participants who entered the study had previously been treated with methotrexate.
GPs with questions or concerns have been advise to contact Pfizer Australia directly on 1800 675 229.
alert arthritis TGA Therapeutic Goods Administration tofacitinib xeljanz
newsGP weekly poll
Would it affect your prescribing if proven obesity management medications were added to the PBS?