News
What is the COVID-19 treatment sotrovimab?
The medication has shown promise in reducing hospitalisation and risk of death, and there is a chance it could be used in primary care. But experts warn the data is limited.
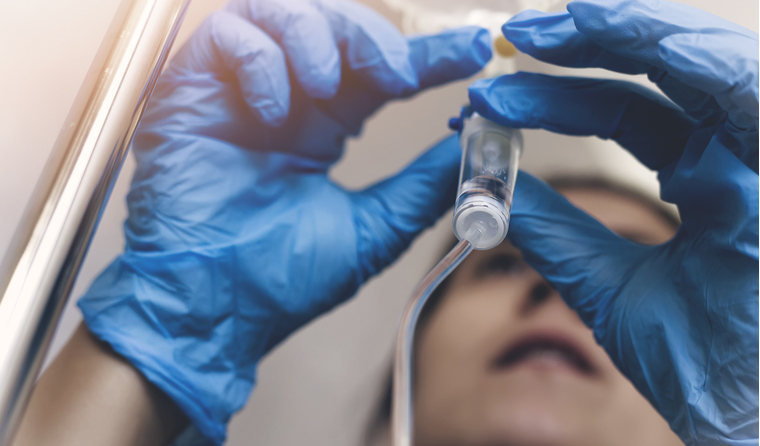 Monoclonal antibodies are designed to mimic natural antibodies produced by the body’s immune system.
Monoclonal antibodies are designed to mimic natural antibodies produced by the body’s immune system.
Sotrovimab, currently approved for use in the US and Europe, is expected to further assist Australia in its efforts to protect people at risk of developing severe COVID-19 and to manage future outbreaks.
Currently under review by the Therapeutic Goods Administration (TGA), the Federal Government has secured an initial shipment of more than 7700 doses for the National Medical Stockpile, with the drug set to be available for use later this year pending regulatory approval.
According to a Government press release, sotrovimab has been shown to reduce hospitalisation or death by 79% in adults with mild-to-moderate COVID-19 who are at high risk of progression to severe disease.
But what exactly is sotrovimab?
Sotrovimab is a novel monoclonal antibody treatment. The laboratory-made proteins are designed to mimic the natural antibodies produced by the immune system when defending itself against diseases.
The monoclonal antibodies are meant to bind to the virus to stop it from entering host cells, and also help to fight off already infected cells.
The treatment is administered intravenously, and a complete treatment cycle requires only one dose.
Will general practice play a role?
Based on past and present experience of managing patients with COVID-19, it is estimated 8–15% of Australians who test positive to the virus would be considered to be at high risk of disease progression and therefore recommended for treatment with sotrovimab.
If the treatment is approved for use in Australia, it will be provided to states and territories from the National Medical Stockpile as needed, and will be made available to the patient free of charge.
However, while the Government has said it will be doctor prescribed and administered in a health facility, it remains unclear whether that will be in primary care.
‘We’re currently working with Australian clinicians and Government to determine the exact treatment settings and to understand how an infusion will best integrate into existing pathways for COVID patients,’ a GSK Australia spokesperson told newsGP.
Professor Mark Morgan, Chair of the RACGP Expert Committee – Quality Care (REC–QC), says clear clinical guidelines will be needed if GPs are to play a role, particularly given the small number of doses that have been secured.
‘The suggestion … is that 8–15% of people that have a positive test might be eligible for this. In a major outbreak that’s going to be huge numbers that have a positive test, way more than the number of doses that have currently been purchased,’ he told newsGP.
‘We’d want some really clear information about when it’s at its most effective, and how to select the right people. And there would need to be potentially some very good connection between the testing of people for SARS-CoV-2 and then the decision to prescribe.
‘You’d want that decision to happen in the most timely way, to get the most effective use out of this treatment.
‘But look, it’s exciting to have something else that can be thought about. It’s not going to be a panacea and I don’t think people should be holding out for it as a treatment.’
What does the evidence say about this potential treatment?
To date, only interim data from phase 3 of the COMET-ICE trial is available. Published in May and yet to be peer-reviewed, the results show the risk of disease progression was reduced by 85%.
While the treatment has been approved by the Europe Medicines Agency and the US Food and Drug Administration for adults and adolescents aged 12 and older and weighing at least 40 kgs, Australia’s Government-funded National COVID-19 Clinical Evidence Taskforce (the Taskforce) recommends the drug should not be used to treat COVID-19 outside of randomised clinical trials.
Taskforce Director Professor Julian Elliott told Nine Newspapers the recommendation is reflective of the limited data available.
‘It was an interim analysis of a single study, with relatively low numbers. The panel decided to give an only-in-research recommendation, pending the availability of more data,’ he said. ‘We don’t base our recommendations on press releases.
‘Other countries have also purchased Sotrovimab. I think the Government trying to get ahead of the curve and purchasing a drug, pending review by the TGA, is reasonable.’
Professor Morgan, who represents the RACGP in the Taskforce’s National Guidelines Leadership Group and is also Co-Chair of the Primary and Chronic Care Expert Panel, believes the monoclonal antibody treatment may well end up being an important part of COVID-19 management. But based on efficacy, says it is ‘nowhere near’ as important as vaccination.
‘The main outcomes of interest, which are to prevent hospitalisation, ventilation and death, both AstraZeneca and Pfizer are more than 90% effective,’ Professor Morgan said.
‘So, vaccinations are both more efficacious and more affordable, and has been tried and tested far more than this treatment, and therefore, we know about its safety.’
Professor Morgan also said the available data provides no insight on whether the treatment is safe and effective during pregnancy or for children.
‘Also, we don’t know whether these neutralising antibodies would affect contagion risk, or what the impacts of current and new variants might be on their effectiveness,’ he said.
During the trial, an intravenous infusion of sotrovimab 500 mg was administered to 291 patients deemed to be at high risk of developing serious COVID-19 and 292 patients were given a placebo, with the treatment started within five days of the patient testing positive to COVID-19 and the onset of symptoms.
High risk was defined as adults aged 55 and over, or adults with at least one of the following risk factors:
- Diabetes requiring medication
- Obesity (body mass index >30 kg/m2)
- Chronic kidney disease (estimated glomerular filtration rate <60 mL/min/1.73 m2)
- Congestive heart failure (New York Heart Association class II or higher)
- Chronic obstructive pulmonary disease
- Moderate-to-severe asthma
The findings showed only three (1%) of those treated with sotrovimab developed severe disease compared to 21 (7%) in the placebo group, and all five patients who were admitted to intensive care, including one who died by day 29, received the placebo.
Meanwhile in the UAE, where sotrovimab was first approved for use in May, the treatment recently demonstrated
100% efficacy in preventing death from COVID-19.
Between 30 June and 13 July, the drug was administered to 6175 patients with mild-to-moderate disease, and within 14 days 97% of recipients had fully recovered with a 99% ICU prevention rate.
Log in below to join the conversation.
comorbidities COVID-19 severe disease sotrovimab treatment
newsGP weekly poll
Would it affect your prescribing if proven obesity management medications were added to the PBS?