Q fever, originally known as ‘query fever’, is a zoonotic disease caused by the Gram-negative, intracellular bacterium Coxiella burnetii.1 It has a low infectious dose (approximately 10–15 organisms for humans),2 and an incubation period of two to three weeks, depending on infectious dose.3 Not all people infected will show clinical symptoms, which can vary from a mild influenza-like illness to an acute condition that requires hospitalisation.4,5
Environment
The main transmission route of C. burnetii to humans is through inhalation of aerosols or dust contaminated by secretions from infected animals, including birth products, faeces and urine. Bacteria can spread over wide areas under the influence of the wind and result in disease outbreaks.6,7 Domestic ruminants (eg cattle, sheep, goats) are considered to be the main source for human infections. Farming and veterinary procedures such as assistance with birthing, slaughtering and butchering provide opportunities for infection. However, C. burnetii has been identified in a diverse range of vertebrates, including other mammals (eg wildlife, domestic and feral dogs and cats), birds and invertebrates (predominantly ticks).8,9 Furthermore, the organism may remain dormant in the soil and dust, and spread by wind, vehicle movements and activities such as lawn mowing.6,7,10
Epidemiology
Q fever infections have been reported from all countries except for New Zealand. In Australia, Q fever is notifiable in all states and territories.11 Notification rates are 6.3 per 100,000 population per annum in Queensland,3.1 per 100,000 per annum in New South Wales, 1.1 per 100,000 per annum in South Australia, and <1.0 per 100,000 per annum in the other states and territories. However, these figures are likely to underestimate the true burden of disease as many cases are undiagnosed.12 Analysis of age and gender distribution shows notifications across the age groups, with males overrepresented in the combined age group 40–69 years (Figure 1).12 In an ongoing prospective cohort study of adults, acute Q fever was found to be highest for adults living on a farm in outer regional or remote areas of New South Wales.13
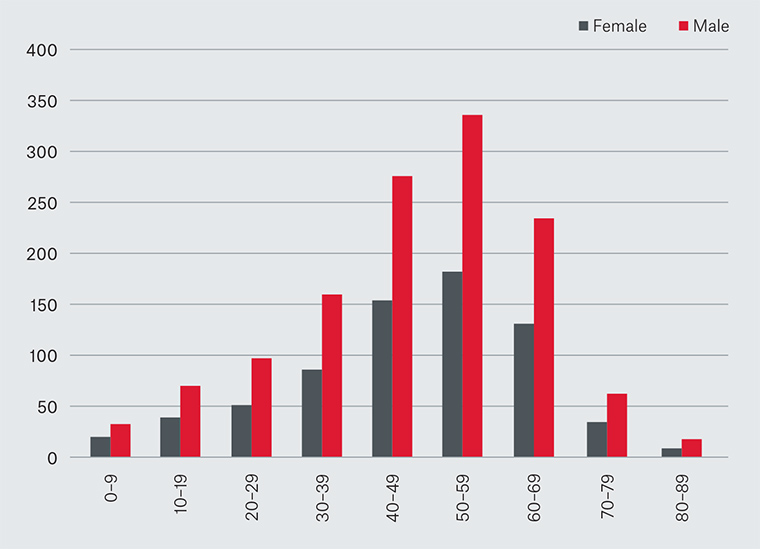
Figure 1. Q fever notifications, by age group and gender, Australia, 2012–16
Seroprevalence studies can provide an indication of the prevalence of C. burnetii infection in a population. In a study of 2122 specimens from a serum bank of samples from a generalised Queensland, Australia, population, 109 specimens were positive. The study found that 5.3% of samples were positive from the rural/remote population. By contrast, in participants from metropolitan Brisbane, an area considered considerably less risky, the rate was similar at 5.0%.14 In a convenience sample from northern New South Wales, another sero-survey found an overall seropositivity rate of 7%, with higher rates in rural areas, compared with urban areas.15 These studies have inherent limitations, biases and confounders, but indicate that Q fever is likely to be more prevalent than the notification rate suggests.
Risk factors
Historically, Q fever has traditionally been associated with the livestock industry (particularly cattle), affecting abattoir workers, farmers, veterinarians, livestock transporters, shearers and wool classers.16,17 Assisting in birthing and slaughtering of animals are particularly high-risk activities.18,19 In a review of Q fever notifications for 2007–14, it was reported that almost two-thirds could be linked to an occupation, animal fluid exposure or environmental exposure known to be associated with Q fever infection.12 However, about half of the notified cases in New South Wales in 2011–15 lacked these risk factors or occupations (HCM Clutterbuck, KE, PDM, K Hope, SM Mor, unpublished data). Recent studies have expanded our understanding of risk determinants to include wildlife carers, cat and dog breeders, people living on livestock transport routes or in proximity to abattoirs and feedlots, people mowing in areas with native animal droppings, following tick bites and urban dwellers exposed to native animals, especially kangaroos and other macropods. Person-to-person spread of Q fever is rare.17 In Australia domestic pets, wild animals and ticks are important reservoirs of C. burnetii, although the exact role they play in the transmission of human disease locally is not well understood.
Pathophysiology
C. burnetii may exist as either a ‘large-cell variant’ (growing form) or as a ‘small-cell variant’ (non-growing, ‘survival’ form). The latter form is highly resistant to desiccation and survives for long periods in the environment. In addition to these morphological forms, C. burnetii displays variations in outer cell surface antigens, which correlate with virulence. Virulent organisms display predominantly phase 1 lipopolysaccharide antigens, whereas avirulent organisms mainly display phase 2 protein antigens. Antibody responses to these antigens are used to distinguish acute, chronic and past Q fever in patients.20
Clinical features and complications
The proportion of symptomatic persons in an outbreak of Q fever has been shown to vary globally from 10% to 85%. Symptoms may vary between individuals, and diagnosis requires laboratory confirmation. Abnormal liver function tests may be evident, although overt jaundice is rare.17 A list of clinical signs and symptoms observed in Australian patients, and the occupations and risk factors that should raise a high clinical index of suspicion for Q fever, are provided in Tables 1 and 2 respectively. The case study below describes a typical Q fever presentation; however, other zoonoses and infections can present similarly. Differential diagnoses of Q fever include Ross River/Barmah Forest viral illness, dengue, Epstein–Barr virus, Legionella infection, psittacosis, leptospirosis, brucellosis, rickettsial infections (ie spotted fever, murine typhus, cat flea typhus), influenza, cytomegalovirus and Mycoplasma pneumoniae pneumonia.
Chronic Q fever
Chronic Q fever is the most serious form of the disease, and can occur months to years after initial illness, even if there is no history of initial illness.21 Routine assessment of valvulopathy has been recommended, as the risk of endocarditis in patients with valvular disease has been estimated at 39%. Diagnosis of chronic Q fever can sometimes be made by real-time, quantitative polymerase chain reaction (qPCR) on the patient’s blood, but expert advice should be sought for interpretation.22
| Table 1. Clinical signs and symptoms of Q fever observed in Australian patients (NSW Health Control guidelines – Q fever) |
| Fever |
| Headache |
| Myalgia and arthralgia |
| Cough |
| Influenza-like illness (rigors, chills, fatigue, sweats) |
| Weight loss |
| Pneumonia |
| Nausea |
| Jaundice (rare) |
| Meningeal signs (rare) |
| Maculopapular rash on trunk (rare) |
| Table 2. Risk factors for considering Q fever vaccination |
| Livestock workers (eg farmers, abattoir workers, shearers, tanners, livestock transporters) |
| Veterinarians, veterinary nurses and veterinary students |
| Wildlife carers, hunters, zoo keepers, animal breeders (eg dogs, cats) |
| Contractors and maintenance workers who visit high-risk areas (eg abattoirs and shearing sheds) |
| People with indirect contact to livestock (eg those living down-wind of livestock transport routes, processing plants, feedlots and abattoirs, ground staff involved in rural mowing) |
Post–Q fever fatigue syndrome
This condition affects 10–15% of patients after acute Q fever. The initial infection may be mild or severe, and patients present with a ‘chronic fatigue-like’ picture. Alcohol intolerance is a commonly reported feature. The only known risk factor for the development of the post–Q fever fatigue is the severity of the initial acute infection.23
Case
A man from northern New South Wales, 54 years of age, presented following 10 days of persistent fevers, chills, rigors, headache and lethargy. He reported drenching night sweats, significantly reduced exercise tolerance, several presyncopal episodes, and four days of non-specific, small-volume, non-bloody diarrhoea.
The man reported having a non-confirmed Q fever infection approximately 17 years prior, and was not vaccinated against Q fever. No other significant medical history was reported.
He lived on a small-acreage farm where he reported frequent direct contact with a small goat herd. This included exposure to the placenta of a goat a fortnight prior to symptom onset. Social history included regular smoking, an active lifestyle and work as a farm-fencing contractor. There was no history of overseas or recent domestic travel.
In the emergency department, the man was clinically diagnosed with Q fever and subsequently hospitalised for 12 days. Initial laboratory tests found elevated C-reactive protein (CRP), low platelets, and deranged liver function tests. The patient was treated with 14 days of doxycycline 100 mg twice daily.
Q fever was confirmed following positive serum PCR for C. burnetii 11 days after the onset of symptoms. However, Q fever immunofluorescence (enzyme immunoassay) serology was negative on the same specimen.
The patient’s admission was complicated with new-onset atrial fibrillation (AF), with a variable rapid ventricular rate fluctuating between 90 and 160 beats per minute. The AF was treated with metoprolol, loaded with digoxin, and apixaban was given for anticoagulation. Digoxin was ceased prior to discharge and the man was referred for an outpatient echocardiogram and cardiac stress test.
Seven weeks after the first negative serology, parallel antibody titres for C. burnetii indicated serological evidence of a recent acute Q fever. Results included:
The case was investigated by the local public health unit. Q fever information was provided to contacts who may have been exposed to the same source as the case. Future Q fever vaccination was contraindicated for the case.
Diagnosis
Because Q fever can be mistaken for other conditions, including other zoonotic diseases (eg leptospirosis, brucellosis), the work-up should be determined by a detailed history, examination and initial screening investigation. A useful algorithm for general practitioners (GPs) that describes the recommended approach to diagnostic tests has been previously reported.24 In addition to the algorithm, the following notes are provided to emphasise aspects assisting diagnosis.
Initial investigations
Full blood count, liver function tests and CRP can give some indication of infection, but are not specific.
Blood culture
Culture for C. burnetii should be avoided (outside of biosafety level 3 laboratory conditions) as it requires specialised culture techniques and, if successful, can be a high risk for laboratory-acquired infections. A blood culture, if requested, should clearly state the possibility of Q fever. Serology, to demonstrate specific antibodies, or through specific bacterial nucleic acid detection are the preferred diagnostic tests.
Nucleic acid testing
Ideally, blood should be collected within one week of disease onset to enable the detection of C. burnetii DNA in blood through Q fever PCR (5 mL EDTA). This test provides the fastest turnaround time to achieve diagnosis, avoiding the need to wait for a convalescent serum ≥2 weeks after. A positive PCR result is confirmatory alongside the clinical findings. The bacteria are rapidly eliminated from the blood stream, so a negative PCR test does not exclude the possibility of Q fever.
Serology
Although a single, high phase 2 IgM antibody titre, in conjunction with a compatible clinical history, is suggestive of acute Q fever infection, it is preferable to demonstrate a fourfold rising antibody titre between an acute blood collection (within seven days of disease onset) and a convalescent specimen collected >7 days after the first collection. A pathology request for phase 1 and phase 2 IgG and IgM C. burnetii serology and titres, preferably by immunofluorescence, is recommended. The convalescent sample should always be tested in parallel (ie in the same test run) with the acute sample so a valid comparison of antibody titres can be achieved. The timing and type of recommended pathology tests are summarised elsewhere.24 Advice from a clinical microbiologist and/or infectious disease physician may be required to assist in the interpretation of the serological results and other aspects of diagnosis.
For suspected chronic Q fever, a request should also be made for phase 1 IgA titres by IFA. In uncomplicated cases, repeat Q fever serology at six months to identify potential progression to chronic disease. For chronic Q fever, expect high phase 1 IgG and IgA titres. These antibody titres do not fall over time, and may increase until effective treatment has begun, after which time titres will eventually fall slowly over many months or years. There is no serological pattern that correlates with the post-Q fever fatigue syndrome.
General/additional pathology testing
A full blood count may show a normal white cell count but, commonly, lymphopenia and thrombocytopaenia are apparent. The liver function profile often shows a raised aspartate aminotransferase and alanine transaminase, but gamma-glutamyl transferase, alkaline phosphatase and bilirubin are usually normal. The CRP may be significantly elevated during the acute phase, but may be normal in chronic Q fever. Rheumatoid factor and other autoantibodies may also be present. It is important to consider transoesophageal echocardiography (TOE) if there is concern about endocarditis; however, TOE can give false negatives when vegetations are not detected. Positron emission tomography or computed tomography may be helpful in chronic Q fever to localise the anatomical site of the chronic/focal infection, and is now used in France.25
Clinical management
Achieving a timely, definitive diagnosis is challenging, but as early treatment is beneficial, empirical antibiotic therapy should be considered if the presentation and clinical history suggest a zoonotic disease. Doxycycline 100 mg twice daily for 14 days is recommended for adults. Referral to an infectious diseases physician for management of children and pregnant women is recommended.
Chronic Q fever requires long-term antimicrobial treatment and specialist input. Chronic Q fever is much more likely in people who have prosthetic cardiac valves, abnormal native valves (eg bicuspid aortic valves), endovascular lesions or are immunosuppressed. Other features of the chronic form include granulomatous hepatitis and osteomyelitis, which is more likely to occur in children.26,27
Public health prevention, infection control and immunisation
Other than surveillance of reported risk factors, there is no specific public health response for a single Q fever case. However, investigation of clusters is valuable as it may identify a common and preventable source. It is important to foster awareness so that people involved in high-risk activities take precautions (eg basic infection control measures, vaccination). It is also important for clinicians to consider Q fever in people with compatible symptoms and/or risk factors so that appropriate treatment is initiated promptly.
Vaccination is the most effective method of prevention; however, good hand hygiene and wearing an N95 mask may be helpful for those who are unvaccinated and working with animals or conducting activities where there are animal droppings and risk of aerosolisation. Exposures recorded in surveillance data indicate that close contact with animal remains, birth products or attendance at birthing are common potential exposure activities.12 Activities such as mowing and slashing where the environment has been contaminated by native animals may also expose a person to aerosolised C. burnetii. Reducing possible exposure pathways by laundering contaminated work clothes away from unvaccinated people, and reducing contact between unvaccinated people and animals in a farm may be helpful.
Vaccination
Vaccination is recommended for all people who are likely to be exposed to C. burnetii as assessed from their environmental, activity or occupational risk (Table 2). The current vaccine is a whole-cell, formalin-inactivated culture, which provides effective immunity but may be reactogenic when administered to people who were previously exposed to the bacteria. Prior to vaccination, a skin test must be performed concurrently with C. burnetii serology testing to test for cellular immunity. Additionally, a medical history aimed at identifying possible previous infection should be collected, and only when all three avenues yield no evidence of prior infection is the person vaccinated.28 The vaccination is safe and effective if used as recommended; local tenderness and erythema are common, and serious adverse events are rare.23 In a review of laboratory-confirmed Q fever notifications for the period 1991–2014 where vaccination status was recorded, only 2% (71/3218) were listed as being previously vaccinated for Q fever, indicating a highly effective vaccine.12 A record of vaccination should be made on the Australian Q Fever Register (www.qfever.org) and a free-text entry made into the Australian Immunisation Register (www.humanservices.gov.au/customer/services/medicare/australian-immunisation-register). A month post-vaccination is required before risk exposures (eg abattoir, cattle yard) is considered.
Issues affecting vaccine uptake have been reviewed for farmers, veterinarians and veterinary assistants.29 Awareness and risk appreciation are often poorly understood by the individual and GP. Additionally, cost and inconvenience to the patient and GP are also factors influencing the uptake of vaccines. The vaccine manufacturer provides some training resources (eg videos) to support GPs acquiring the knowledge and skills to effectively and safely immunise against Q fever (www.qfever.org). The vaccine is currently not registered for use in children <15 years of age, but is currently being trialled in children 10–14 years of age.
Summary of important points
Q fever is the most commonly notified zoonotic disease in Australia, with the majority of cases reported from northern New South Wales and southern Queensland. It can affect all persons, but most notifications are from middle-aged males.
Q fever cases present with a range of symptoms, often as undifferentiated febrile illness that cannot be distinguished from a number of other conditions. It is most promptly diagnosed through PCR or later through serology, especially via rising antibody titres in paired sera.
C. burnetii responds to antibiotic therapy: doxycycline for adults. However, chronic Q fever, which involves vascular infections and endocarditis, hepatitis or osteomyelitis (in children), provides treatment dilemmas and requires infectious diseases specialist intervention.
Treatment of post-Q fever fatigue syndrome is very challenging, and psychologist involvement, along with gentle, supervised exercises, may offer some relief to the patient.
Although the most common mode of transmission is through exposure to livestock, many other animals carry the bacteria. C.burnetii can remain in the environment for long periods, resulting in transmission through inhalation of dust particles, which can provide obscure exposure mechanisms. An effective vaccine is available in Australia; however, due to its reactogenic nature, a careful pre-vaccination assessment (involving skin test, serology and clinical history) is required. Additionally, cost, inconvenience due to the two-step vaccination process, access to vaccination and safety concerns discourage uptake. The main issues for patients and clinicians are a lack of disease awareness and a misunderstanding of the breadth of risk factors associated with Q fever.