Transcranial magnetic stimulation (TMS) is a means of non-invasively stimulating the cerebral cortex. It is an application of electromagnetism, and has a place in diagnostic neurophysiology and the treatment of some neurological and psychiatric disorders.
Here, we describe TMS as useful in the treatment of major depressive disorder (MDD) that is otherwise resistant to treatment. It is currently reserved for patients who have failed to respond to at least one course of an antidepressant medication administered at a sufficient dose for a sufficient period – that is, patients with treatment-resistant depression (TRD).1 Currently, TMS is used predominantly in adults, and is effective in young and old alike.2 This treatment has had limited availability in Australia in academic, private and public practices for two decades.
History and physiology
Electroconvulsive therapy (ECT) was first performed in Rome in the 1930s. It has the strongest available antidepressant effect,3 but the requirement for general anaesthesia and induction of seizure, and the side effects (including temporary memory problems) make it unacceptable or inappropriate for some patients.
TMS became possible in 1985.4 It depends on the following. When electricity passes along a wire, a magnetic field simultaneously extends at right angles around the wire. Relevant principles are illustrated by the transformer – a device in which a coil of wire in one circuit is placed close to a coil of wire in a second circuit. When a current is passed through the first circuit, a current also passes through the second (to which it is not electrically connected) – the magnetic field of the first coil reaches the second coil, and produces the electric current in that circuit.
The second structure need not be a metal coil; the magnetic field emanating from a coil will induce a small current in any nearby conductor, including a human brain.
Unlike electricity (in ECT), magnetic fields (such as those of TMS) pass directly through the skull (a non-conductor of electricity), with little loss of energy. In the same way, the magnetic field of a magnet held under a wooden table (also a non-conductor of electricity) easily reaches a paperclip on the tabletop. Thus, TMS‑generated cerebral electrical activity is tightly focused and can be localised; there is no seizure, no need for anaesthetic, and no memory problems.5
The first TMS machines provided only single pulses, followed by a delay during which electrical charge re-accumulated. However, the development of the thyristor (a solid-state semiconductor device) enabled the building of machines, in the mid-1990s, capable of delivering repetitive TMS (rTMS) pulses at up to 20 Hz. Physiological studies have demonstrated that low-frequency (<1 Hz) rTMS decreases the activity of neural tissue,6 while high-frequency (>1 Hz) TMS increases neural activity.7 Neuroimaging studies indicated that in MDD, the left dorsolateral prefrontal cortex (DLPFC) is underactive when compared with the right DLPFC.8 Thus, two treatment methods emerged: high frequency to the left DLPFC and low frequency to the right DLPFC.
A successful placebo-controlled, randomised study of high frequency rTMS in TRD was reported in 1997.9 Success with low frequency rTMS was reported in 1999.10
Imaging
Contemporary TMS imaging studies have explored connectivity – defined as a measure of the extent to which the components (nodes) of a network are connected to one another, and the ease (speed) with which they can ‘converse’ – of three recently described networks: the default mode network, central executive network and the salience network. All have nodes in the prefrontal cortex (the site of TMS stimulation), and connections to subcortical nodes. Depression is associated with dysfunctional connectivity in these networks, and successful rTMS treatment is associated with normalisation of these abnormalities.11–14
Current evidence
Four recent publications warrant consideration:
-
position statement of the Royal Australian and New Zealand College of Psychiatrists (RANZCP)15
-
guidelines prepared by European experts16
-
recommendations of the Clinical TMS Society17
-
guidelines of the Canadian Network for Mood and Anxiety Treatments.18
The Canadian authors18 found that rTMS (both high and low frequency) is suitable as first-line treatment for treatment-resistant MDD, grading the evidence as ‘Level I’. The European authors16 gave rTMS (both high and low frequency) a ‘Level A recommendation’ and reported ‘definite efficacy’. The Clinical TMS Society17 reached similar conclusions, and the RANZCP15 (using earlier information) found ‘robust evidence’ of efficacy in MDD.
The above groups found that rTMS is a less effective antidepressant than ECT, although the difference may be slight.16 However, rTMS is not complicated by the side effects that may make ECT less acceptable.
Patient selection
rTMS is currently reserved for adults with well-documented TRD, which can be unipolar or bipolar. Patients frequently continue antidepressants and mood stabilisers when receiving TMS; however, there is need for compliance with local treatment centre protocols. rTMS should be considered before advancing to ECT (when available) in all but the most self-dangerous cases.16
The contraindications to treatment vary, depending on local treatment centre protocols. The following is a general guide to contraindications:
-
a personal history of seizure; in the case of an individual who does not have epilepsy, but has a strong family history, caution is recommended
-
pregnancy
-
any condition that may lower the seizure threshold (eg significant past head injury, current alcohol withdrawal)
-
intracranial metal objects and devices
-
cardiac pacemakers – need to be discussed with a cardiologist.
Treatment
The patient is seated comfortably and the coil is secured (touching the hair/scalp) by an adjustable arm (Figure 1). A standard daily stimulation protocol is pulses at 10 Hz in four-second trains, with 26 seconds of rest between trains, and a total of 75 trains. Thus, 3000 stimuli are delivered in a little over half an hour. A course of treatment is 20 daily sessions over 22 days.
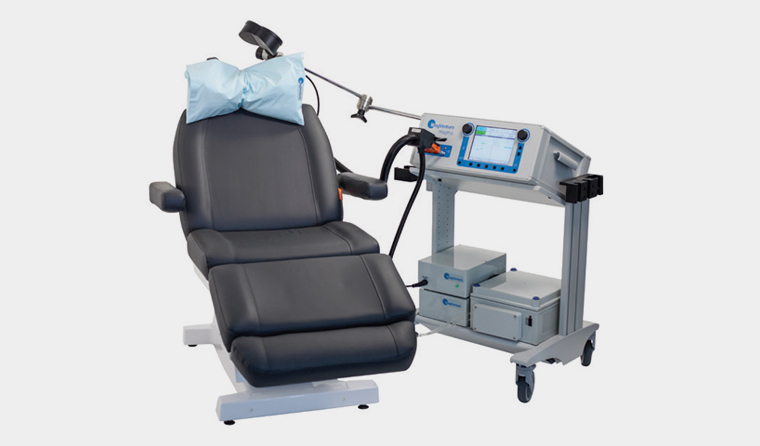 Figure 1. rTMS device with cable and coil secured by mechanical arm
Figure 1. rTMS device with cable and coil secured by mechanical arm
Reproduced with permission from Sonoray Pty Ltd
A suitably trained medical practitioner determines the appropriate stimulation details at the first treatment. For subsequent treatments, a trained health professional (usually a nurse) positions the patient and the coil, and remains in the facility throughout the procedure.
Patients who come to rTMS have failed pharmacological therapy and, accordingly, have TRD, a condition in which relapse is a common feature.1 Relapse in patients with TRD who are treated with rTMS may be considered as much a function of the difficult-to-manage disorder as a failure of the last treatment. Following rTMS-induced remission, the mean time to relapse is five months,19 which is similar to that of ECT.
Adverse effects
Adverse effects are generally trivial rather than serious and there are no long-term undesirable effects. Magnetic pulses may cause scalp discomfort, which usually becomes less annoying for the patient as the course progresses. Some patients develop headache, which responds to simple analgesia – long-term headaches are not a consequence. Anecdotal evidence suggests patients with bipolar disorder can begin to experience elevated mood, but they settle with treatment pauses. Seizure has been reported; however, the incidence (~0.01–0.1%) is less than when commencing an antidepressant, and only slightly greater than the spontaneous incidence in the general population.18
Naturalistic study outcomes
Given that patients treated with rTMS have TRD, improvement will be difficult to achieve. A meta-analysis of naturalistic high-frequency rTMS to the left DLPFC found 25–35% remission;20 another meta-analysis that considered low-frequency rTMS to the right DLPFC found 35% remission.21
Cost-effectiveness
Cost-effectiveness studies have compared TMS and ECT. Given TMS involves more treatments (approximately 20, compared with approximately six), ECT might be expected to have unanimous advantage. However, the first study, performed in the US, concluded that with wide availability TMS would offer an economic advantage and an increase in quality-adjusted life years.22 One European study found that, overall, there were no treatment cost differences,23 and the most recent study, performed in Singapore, found TMS more cost-effective than ECT over one year.24
A recent study in the US reported that for patients newly diagnosed with MDD who have failed a single antidepressant drug trial, there would be cost saving and greater clinical efficacy if the patient was treated thereafter with TMS, for life.25 Thus, it can be expected that the future treatment of TRD will involve TMS for at least a proportion of patients.
Other conditions treated
At this time, we recommend TMS only for the treatment of TRD. Within psychiatry, research continues with low frequency rTMS to the left temporal region as a potential treatment for auditory hallucinations. In neurology, rTMS may have a place in rehabilitation following cerebrovascular accident.26 Some benefit has also been claimed in the treatment of tinnitus, Parkinson’s disease and chronic pain.27–29 TRD is the only condition for which unequivocal evidence is available.
Availability
rTMS is available in Australia in various academic, private and occasional public settings, but is not widely available. rTMS does not yet have a Medicare Benefits Schedule item number, but should that happen, availability is expected to increase.
Conclusion
rTMS uses electromagnetism to place small electric currents in localised regions of the cortex. It is effective in the management of TRD, in both placebo-controlled and naturalistic studies. TRD is major treatment challenge and, thus, rTMS is an important medical advance.
rTMS does not cause seizure or the memory problems associated with ECT. The side-effect profile is generally trivial and there are no long-term side effects. rTMS-induced remission is associated with the normalisation of cortical–subcortical connectivity abnormalities. Availability in Australia has been limited; however, services continue to be established and availability is increasing.