Case
A woman aged 68 years with a history of hypertension presented to her general practitioner (GP) with a one-day history of a painful rash on the left side of her forehead. The eruption was preceded by a two-day history of burning pain. She had no ocular symptoms. Clinical examination showed a unilateral dermatomal vesicular rash consistent with a left-sided varicella-zoster virus (VZV) infection of the first branch of the trigeminal nerve (shingles). The patient did not smoke and had an alcohol intake of approximately two standard drinks per day and two alcohol-free days per week. Incidentally, she was also scheduled to review the results of her annual blood examination results taken five days earlier. The pathology service provider used routinely includes lactate dehydrogenase (LDH) in requests for liver function tests (LFTs). Her blood test results (Table 1) showed mild anaemia, reticulocytosis, macrocytosis, hyperbilirubinaemia and elevated LDH. Red cell agglutination and polychromasia were reported on the blood film (Figure 1).
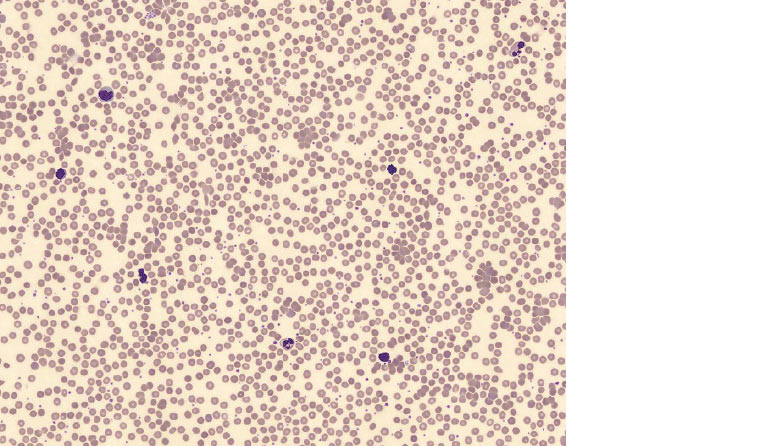
Figure 1. Red cell agglutination and polychromasia on peripheral blood film
Question 1
What important underlying conditions should be considered in this case?
Question 2
What further information should be sought in history and examination?
Answer 1
In Australia, most cases of shingles occur in immunocompetent adults with no identifiable trigger1 other than increasing age.2 Occasionally, the reactivation of VZV is triggered by an underlying haematological malignancy or an immunocompromised state arising from immunosuppressive therapy following organ/stem cell transplantation or infection with human immunodeficiency virus.1,2 Importantly, the constellation of abnormalities noted on the blood results should prompt the clinician to consider autoimmune haemolytic anaemia (AIHA) due to cold agglutinins (CAs).3
CAs are autoantibodies that optimally agglutinate red cells at temperatures of 3–4 °C, although they are capable of agglutination at higher temperatures.4 The presence of CAs does not always have clinical significance,3,5 but it may rarely cause complement-mediated AIHA in the forms of idiopathic CA disease (CAD) or secondary CA syndrome (CAS) due to acute infections, autoimmune disease or lymphoproliferative disorders (LPDs).3,5,6 Thus, the clinical conundrum in this case is whether this was transient post-infectious CAS, or whether an underlying LPD was the common trigger for VZV reactivation and/or CAS. Infectious exacerbation of undiagnosed CAD also merits a consideration because CAD predominantly affects women in their seventh decade of life, with a reported median age at symptom onset of 67 years.7
Answer 2
CAD generally causes chronic mild-to-moderate anaemia.6 However, life-threatening haemolysis has been reported in CAS8 and during exacerbations of CAD from cold exposure,6 acute febrile illness, trauma or surgery.5 Therefore, evaluation for haemodynamic compromise and secondary causes is important.
Case continued
At this visit, other than symptoms of her shingles, the patient was well. She had no constitutional symptoms. She did not have any hair loss, rashes, mucosal ulcers, joint pains, acrocyanosis or cold-induced symptoms. She was haemodynamically stable with no palpable lymphadenopathy and no hepatosplenomegaly.
Question 3
What further tests should be considered in this case?
Answer 3
Diagnosis of typical herpes zoster can be made on clinical grounds.2,9 However, diagnosis of CAD requires evidence of chronic haemolysis, CA titre ≥64 at 4 °C and a positive direct antiglobulin test (DAT) for anti-C3 with negative anti-immunoglobulin G (anti-IgG).4,5,10 Evaluation for secondary causes5 is also recommended, especially in adults >60 years of age.3
Case continued
In consultation with haematologists, investigations for multiple myeloma, autoimmune and haemolytic screens, peripheral blood surface markers, haematinic profile, DAT and CA titre were requested. Serial full blood examination (FBE) and LFTs were requested to track ongoing haemolysis.
The patient’s DAT was positive for anti-C3 and negative for anti-IgG. She had a positive CA (anti-I specific) titre of 64 at 4 °C. No immunophenotypic abnormality was detected on cell surface markers. The antinuclear antibody screen was negative. Her serum protein electrophoresis, iron status, vitamin B12 and folate levels were normal.
Follow-up results showed normalisation of CA titre, haemoglobin, platelet count and LDH but persistent red cell agglutination beyond three months (Table 1). This prompted a review of her previous blood results, which showed reports of red cell agglutination but normal FBE indices and no evidence of haemolysis over the preceding four years. This suggests pre-existing but benign CA, with the transient haemolysis being most likely triggered by an intercurrent illness – shingles. Cases of varicella-associated CAS reported in the literature have been in the context of chickenpox4 rather than shingles.
| Table 1. Results of serial full blood examination, renal function test, liver function tests and cold agglutinin titre |
| |
One year
prior |
At
presentation |
Two weeks later |
Three weeks later |
Four weeks later |
Three months later |
Four months later |
Haemoglobin
(g/L; ref. 110–160 g/L) |
120 |
100 |
106 |
N/A |
107 |
110 |
114 |
|
Red cell count
(1012/L; ref. 3.7–5.4 × 1012/L)
|
3.8 |
3.1 |
3.2 |
N/A |
3.4 |
3.4 |
3.6 |
|
Reticulocyte count
(109/L; ref. 25–120 × 109/L)
|
N/A |
211 |
N/A |
191 |
149 |
N/A |
161 |
|
Mean corpuscular volume
(fL; ref. 80–100 fL)
|
91 |
101 |
96 |
N/A |
103 |
100 |
100 |
|
White cell count
(109/L; ref. 4.0–11.0 × 109/L)
|
8.4 |
7.4 |
11 |
N/A |
8.4 |
9.8 |
8.6 |
Platelet
(109/L; ref. 150–450 × 109/L) |
410 |
426 |
465 |
N/A |
390 |
403 |
397 |
|
Lactate dehydrogenase
(U/L; ref. 120–250 U/L)
|
205 |
254 |
260 |
N/A |
N/A |
221 |
218 |
Total bilirubin
(µmol/L; ref. <16 µmol/L) |
12 |
26 |
40 |
27 |
37 |
25 |
38 |
| CA titre (°C; ref. 4 °C) |
N/A |
N/A |
N/A |
64 |
<32 |
N/A |
N/A |
| Red cell agglutination on blood film |
Present |
Present |
Present |
Present |
Present |
Present |
Present |
Sodium
(mmol/L; ref. 135–145 mmol/L) |
N/A |
136 |
N/A |
N/A |
N/A |
N/A |
N/A |
|
Potassium
(mmol/L; ref. 3.5–5.5 mmol/L)
|
N/A |
4.3 |
N/A |
N/A |
N/A |
N/A |
N/A |
Urea
(mmol/L; ref. 3.0–8.5 mmol/L) |
N/A |
4.3 |
N/A |
N/A |
N/A |
N/A |
N/A |
Creatinine
(µmol/L; ref. 45–85 µmol/L) |
N/A |
48 |
N/A |
N/A |
N/A |
N/A |
N/A |
| CA, cold agglutinin; N/A, not available; ref, reference |
Question 4
How would you manage this patient?
Answer 4
Management of CAS is guided by severity of symptoms and any identified aetiology.6 Management of mild cases includes education on cold avoidance and prompt treatment of febrile illnesses.3,6 Patients with severe haemolysis and haemodynamic compromise will require supportive transfusion3,6 and non-GP specialist input. Patients with confirmed/suspected serious underlying aetiology should be managed in conjunction with non-GP specialists.
Conclusion
The patient’s shingles was managed with a seven-day course of oral famciclovir with relevant safety netting advice. General advice about the diagnosis and potential triggers for future exacerbations was provided. The patient’s immunisation record showed that recommended vaccinations were up to date. Her shingles resolved without further complication and she remained clinically well at her follow-up at four months.