Case
A Caucasian woman aged 65 years presented with painful mouth ulcers (Figure 1A), conjunctivitis (Figure 1B) and a new blistering eruption on the central chest (Figure 1C) and forearm (Figure 1D) 10 days after commencing topical imiquimod 5% cream to treat biopsy-proven superficial basal cell carcinoma (BCC) of the left forearm and hip. Regular medications included rosuvastatin, citalopram and paracetamol, none of which were new. There were no known allergies.
On examination, the patient was febrile (temperature 39.3 °C) and had tachycardia (heart rate 120 beats per minute). There were multiple eroded lesions on the central chest. Sites of treatment with imiquimod showed an expected level of local inflammation and crusting. There was marked conjunctival injection but no objective visual changes. There was significant ulceration of the lower labial mucosa and the hard palate.
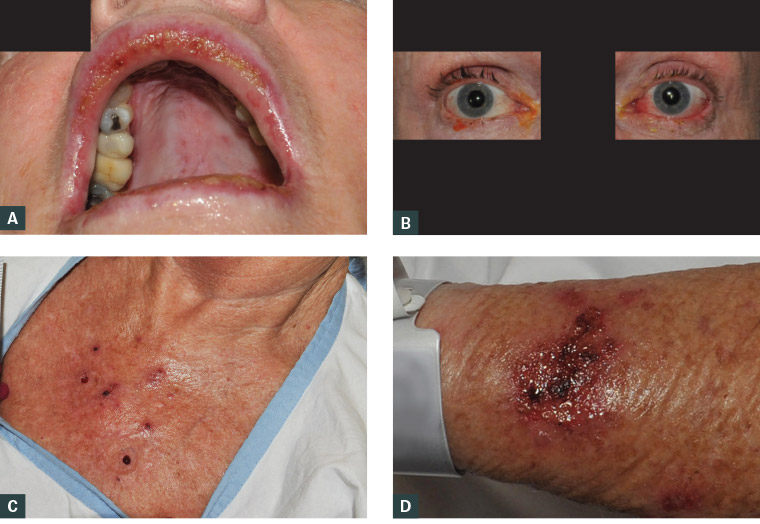
Figure 1. Findings on patient examination
A. Oral mucosal ulceration; B. Conjunctivitis; C. Multiple blistering lesions on the central chest; D. Expected cutaneous reaction to topical imiquimod cream
Question 1
What differential diagnosis should be considered?
Question 2
How is this condition diagnosed?
Question 3
What is the appropriate management of this condition?
Answer 1
With such a clear temporal relationship with the commencement of a new medication, significant mucosal involvement and systemic symptoms, a severe cutaneous drug reaction is the primary differential diagnosis. In this case, the spectrum includes Stevens–Johnson syndrome (SJS) and toxic epidermal necrolysis (TEN). Other important differentials include erythema multiforme, a fixed drug eruption, cutaneous lupus and immunobullous disorders (particularly pemphigus vulgaris; other immunobullous disorders would typically show intact blisters). An infective precipitant should be ruled out, particularly herpes simplex and varicella zoster virus (VZV).
Answer 2
Suspected drug-related exanthems without potential for systemic compromise could be investigated initially by the general practitioner (GP). In contrast, suspected drug-related eruptions with blistering, especially those with mucosal involvement, should be referred immediately to hospital for investigation because of the risk of progression and systemic compromise from skin barrier dysfunction (eg temperature dysregulation, infection, eye involvement and reduced oral intake due to mucosal involvement).
Suspected causative agents should be immediately ceased. Cutaneous biopsy of lesional skin for histological examination and of perilesional skin for direct immunofluorescence is indicated for suspected cutaneous drug reactions.1 Full blood examination, liver and renal function tests, swabs for viral triggers and a general septic screen (including a chest X-ray and urine culture) should be performed at the outset. Such investigations are usually performed in hospital but can be commenced in the general practice setting if clinically appropriate.1
Answer 3
Patients with SJS/TEN should be transferred to a tertiary centre for close observation with access to a burns unit/intensive care. All cases of SJS/TEN should have ophthalmology review because of the risk of synechiae and corneal scarring.2,3
Case continued
Topical imiquimod was ceased by the GP, and the patient was referred to hospital. Intensive skin, mouth and eye care was implemented.
Histological examination showed focal parakeratosis overlying an epidermis with abundant apoptotic keratinocytes. There was full-thickness epidermal necrosis. There was a subepidermal split leading to bulla formation. The superficial dermis contained pigment-laden macrophages and a mixed inflammatory infiltrate (Figure 2). The features were of an interface-obscuring lichenoid reaction, and the differential diagnosis included erythema multiforme, SJS and TEN.
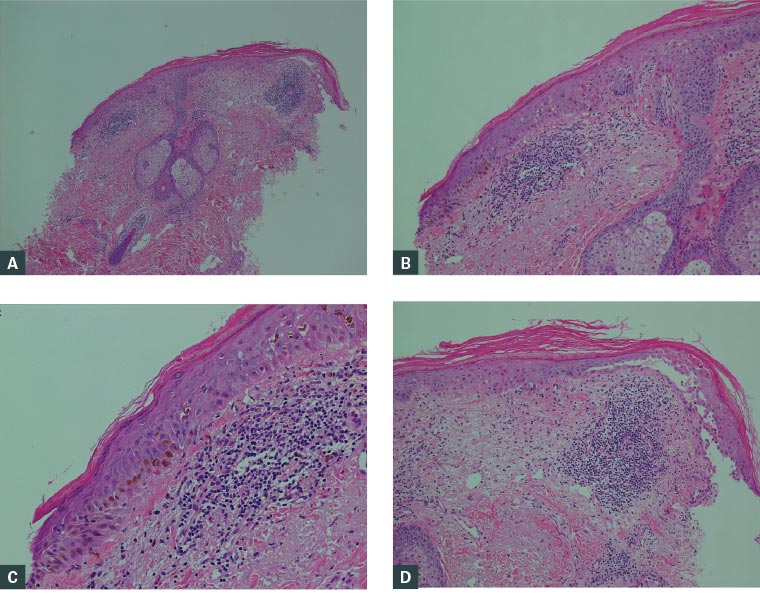
Figure 2. Sections of the punch biopsy, showing a dense lymphocytic infiltrate within the dermis with scattered neutrophils present
A. & B. Parakeratosis and apoptotic keratinocytes within the epidermis; C. Pigment incontinence; D. Subepidermal bulla formation
Full blood examination and electrolytes/liver function were unremarkable. Serial blood cultures were negative. Urine microscopy and culture was clear. Chest X-ray was clear. Viral serology was negative for VZV, cytomegalovirus and Epstein–Barr virus. Mycoplasma pneumoniae antibody was negative. Serial swabs from the eye, mouth and skin returned negative polymerase chain reaction results for herpes simplex virus types 1 and 2, and VZV.
Flexible laryngoscopy showed extension of mucosal involvement to the larynx.
Considering the patient’s extensive mucosal involvement, with <10% of the body surface area affected, SJS was the diagnosis.
The patient was commenced on a pureed, high-energy and high-protein diet. Input was sought from dermatology; ear, nose and throat specialists; ophthalmology; general medicine and allied health specialists.
Question 4
What is the prognosis for this condition?
Answer 4
SCORTEN and, more recently, ABCD-10 (age, bicarbonate, cancer, dialysis, 10% body surface area) are illness severity scores for the prediction of mortality in SJS/TEN. Using the SCORTEN scale, factors associated with increased mortality include higher age (>40 years), tachycardia, increased serum bicarbonate/urea/glucose level at the time of diagnosis, presence of malignancy and a large surface area of skin detachment (>10%). A SCORTEN score of ≥5 is associated with >90% mortality.4
Case continued
With a SCORTEN score of 2 (age >40 years, tachycardia), this patient’s predicted mortality rate was 12.1%. Fortunately, she had complete symptomatic improvement and was discharged after seven days.
Question 5
Is topical imiquimod known to cause severe cutaneous drug reactions?
Answer 5
Topical imiquimod is generally well tolerated for treatment of superficial BCC, solar keratosis and warts. Local skin reactions are an expected part of treatment, and patients can experience systemic symptoms such as headache, fatigue, myalgia and nausea.5 Severe cutaneous reactions such as erythema multiforme, SJS and TEN have been reported and are listed as a potential adverse effect within the extended product information for imiquimod.5,6 There are several case reports regarding the occurrence of erythema multiforme secondary to topical imiquimod.7,8 There are limited case reports of SJS/TEN following use of topical imiquimod.9
Key points
- Topically administered medications can cause cutaneous and systemic reactions.
- Patients with severe cutaneous drug reactions such as SJS/TEN can present in the general practice setting.
- Suspected SJS/TEN should be referred to a tertiary centre for work-up and intensive monitoring/care because of high mortality rates.
- Less worrying suspected drug eruptions (ie those without systemic compromise or blistering) can be investigated in the general practice setting with cutaneous biopsy and other basic screening investigations (full blood examination, liver and renal function tests).
- SJS/TEN is a rare but significant side effect of which practitioners should be aware when prescribing medications, including topical varieties.