Heavy menstrual bleeding (HMB) has been defined as ‘excessive menstrual blood loss which interferes with the woman’s physical, emotional, social and material quality of life, and which can occur alone or in combination with other symptoms’.1 There is evidence from the UK that HMB affects approximately 25% of women of reproductive age.2 However, there has been little published on Australian general practitioner (GP) management of HMB.
The Australian Commission on Safety and Quality in Health Care published the HMB clinical care standard in October 2017.3 The Standard includes eight quality statements describing the clinical care a patient experiencing HMB should be offered, six of which relate to GPs (Box 1). The aim of this study was to examine the GP management rate of HMB from 2000 to 2016, and to assess their management actions for HMB from 2008 to 2016 in light of the recommendations in the recent clinical care standard.
| Box 1. Heavy menstrual bleeding clinical care standard quality statements |
1. Assessment and diagnosis. The initial assessment of a woman presenting with heavy menstrual bleeding includes a detailed medical history, assessment of impact on quality of life, a physical examination and exclusion of pregnancy, iron deficiency and anaemia. Further investigations are based on the initial assessment.
2. Informed choice and shared decision making. A woman with heavy menstrual bleeding is provided with consumer-focused information about her treatment options and their potential benefits and risks. She is asked about her preferences in order to support shared decision making for her clinical situation.
3. Initial treatment is pharmaceutical. A woman with heavy menstrual bleeding is offered pharmaceutical treatment, taking into account evidence-based guidelines, her individual needs and any associated symptoms. Initial treatment is provided to a woman who is undergoing further investigations to exclude malignancy and significant pathology
4. Quality ultrasound. A woman having an ultrasound to investigate the cause of her heavy menstrual bleeding has a pelvic (preferably transvaginal) ultrasound, which assesses endometrial thickness and uterine morphology in days 5–10 of her menstrual cycle.
5. Intrauterine hormonal devices. When pharmaceutical treatment is being considered, the woman is offered the levonorgestrel intrauterine system if clinically appropriate, as it is the most effective medical option for managing heavy menstrual bleeding.
6. Specialist referral. A woman with heavy menstrual bleeding is referred for early specialist review when there is a suspicion of malignancy or other significant pathology based on clinical assessment or ultrasound. Referral is also arranged for a woman who has not responded after six months of medical treatment. |
| Reproduced with permission from Australian Commission on Safety and Quality in Health Care, Heavy menstrual bleeding clinical care standard, Sydney, NSW: Australian Commission on Safety and Quality in Health Care, 2017. |
Methods
An analysis of encounters with female patients of reproductive age (defined as 12–54 years, inclusive) from the Bettering the Evaluation and Care of Health (BEACH) program between April 2000 and March 2016, and an analysis of management in the most recent half of the study (2008–16) were conducted. BEACH was a continuous, national, cross-sectional study of Australian GP clinical activity. Its methods have been described in detail elsewhere.4 In summary, each year a new random sample of approximately 1000 GPs each recorded details for 100 consecutive encounters with unidentified, consenting patients, on structured paper forms.
All aspects of the GP–patient encounter were recorded. Patient demographic data included date of birth; sex; and whether the patient identified as Aboriginal or Torres Strait Islander, was from a non-English speaking background (defined as primarily speaks a language other than English at home), held a Commonwealth healthcare card (CHC) or was new to the practice. GPs could record up to four problems managed at each encounter, and whether each was a new problem for this patient (new) or a pre-existing problem seen before by a medical practitioner (old). Management actions were directly linked to each problem by the GP and included medications prescribed, supplied or advised for over-the-counter purchase, non-pharmacological treatments (eg counselling, advice), pathology and imaging requests, and referrals. Demographic data on participating GPs were also collected.
The encounter information was secondarily entered by trained clinical coders. All problems managed, non-pharmacological treatments, referrals, and pathology and imaging were coded using the International Classification of Primary Care Version 2 (ICPC-2) PLUS, a GP terminology classified to ICPC-2.5,6 HMB was defined as ICPC-2 rubric X06: Menstruation excessive. Medications were coded using the Coding Atlas for Pharmaceutical Substances and classified to the Anatomical Therapeutic Chemical classification.7,8 The definitions used for medications are in Appendix 1.
The authors calculated robust 95% confidence intervals (CIs) using survey procedures in SAS 9.4, which accounted for the cluster sample design. Statistical significance of differences was determined by non-overlapping 95% CIs. This estimate of difference is more conservative than the usual 5% level, reducing the risk of type I errors, while increasing the risk of type II errors.9
The BEACH program received ethics approval from the Human Research Ethics Committee of the University of Sydney (reference no.: 2012/130) and the Australian Institute of Health and Welfare (AIHW) Ethics Committee for the years they collaborated (2006–11).
Results
From April 2000 to March 2016, 15,659 GPs recorded 461,551 encounters with female patients aged 12–54 years. HMB was managed 3264 times at a rate of 7.1/1000 encounters, significantly increasing from 6.3/1000 encounters in 2000–04 to 8.1/1000 in 2012–16 (Figure 1).
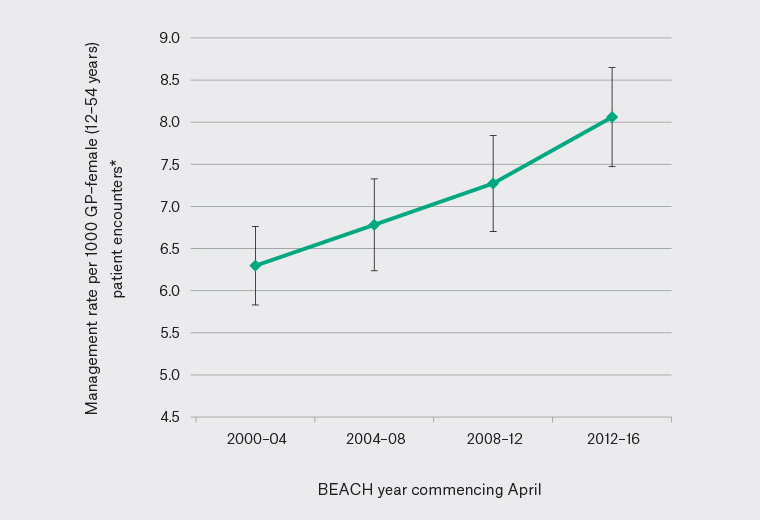
Figure 1. Management of heavy menstrual bleeding per 1000 general practitioner (GP) encounters with female patients aged 12–54 years in Australian general practice (April 2000–March 2016)
*Error bars indicate 95% confidence intervals. BEACH, Bettering the Evaluation and Care of Health
The highest management rate was among women aged 35–44 years (10/1000 encounters), followed by 45–54 years (8.8/1000), and then girls aged 12–17 years (7.1/1000). The management rate was significantly higher among patients without a CHC compared with card holders (7.4, compared with 6.6) and among patients seen previously at the practice compared with those who were new (7.2, compared with 5.9). There were no significant differences in management rates for Aboriginal and Torres Strait Islander peoples (compared with non-Indigenous people) or those with a non-English speaking (compared with an English speaking) background. Female GPs managed HMB at more than double the rate compared with male GPs. There was no statistical difference in management frequency between GP age groups when GP sex was taken into account (Table 1).
| Table 1. Patient and general practitioner characteristic-specific management rate of HMB per 1000 encounters |
| |
Number of encounters
(n = 461,551)
n (%) |
Number of HMB problems managed
(n= 3,264)
n (%) |
Variable-specific HMB management rate per 1,000 encounters (95% CI) |
| Patient age (years) |
|
|
|
| 12–17 |
31,501 (6.8%) |
225 (6.9%) |
7.1 (6.2, 8.1) |
| 18–24 |
69,961 (15.2%) |
281 (8.6%) |
4.0 (3.5, 4.5) |
| 25–34 |
116,717 (25.3%) |
481 (14.7%) |
4.1 (3.7, 4.5) |
| 35–44 |
117,121 (25.4%) |
1,170 (35.8%) |
10.0 (9.4, 10.6) |
| 45–54 |
126,251 (27.4%) |
1,107 (33.9%) |
8.8 (8.2, 9.3) |
| Indigenous status (missing n) |
(80,486) |
(505) |
|
| Aboriginal and Torres Strait Islander |
9107 (2.4%) |
68 (2.5%) |
7.5 (5.6, 9.3) |
| Non-Indigenous |
371,958 (97.6%) |
2,691 (97.5%) |
7.2 (6.9, 7.5) |
| Language background (missing n) |
(50,130) |
(329) |
|
| Non-English speaking |
35,209 (8.6%) |
245 (8.3%) |
7.0 (6.0, 7.9) |
| English speaking |
376,212 (91.4%) |
2,690 (91.7%) |
7.2 (6.9, 7.4) |
| Commonwealth healthcare card (missing n) |
(63,871) |
(406) |
|
| Patient has card |
121,258 (30.5%) |
800 (28.0%) |
6.6 (6.1, 7.1) |
| Patient does not have card |
276,422 (69.5%) |
2,058 (72.0%) |
7.4 (7.1, 7.8) |
| Status to practice (missing n) |
(6182) |
(45) |
|
| New patient |
46,541 (10.2%) |
276 (8.6%) |
5.9 (5.2, 6.7) |
| Existing patient |
408,828 (89.8%) |
2943 (91.4%) |
7.2 (6.9, 7.5) |
| GP age and sex (n = 15,659) |
|
|
|
| Male – total (n = 9679, 62.2%) |
228,893 (49.6%) |
1014 (31.1%) |
4.4 (4.1, 4.7) |
| Male <35 years (n = 428, 2.8%) |
12,534 (2.7%) |
70 (2.2%) |
5.6 (4.1, 7.0) |
| Male 35–44 years (n = 1741, 11.2%) |
45,993 (10.0%) |
192 (5.9%) |
4.2 (3.6, 4.8) |
| Male 45–54 years (n = 3023, 19.4%) |
74,092 (16.1%) |
322 (9.9%) |
4.3 (3.8, 4.8) |
| Male >55 years (n = 4487, 28.8%) |
94,839 (20.7%) |
425 (13.1%) |
4.5 (4.0, 4.9) |
| Female – total (n = 5887, 37.8%) |
232,658 (50.4%) |
2250 (68.9%) |
9.7 (9.2, 10.1) |
| Female <35 years (n = 627, 4.0%) |
27,534 (6.0%) |
247 (7.66%) |
9.0 (7.7, 10.2) |
| Female 35–44 years (n = 1638, 10.5%) |
69,895 (15.2%) |
709 (21.9%) |
10.1 (9.3, 11.0) |
| Female 45–54 years (n = 2210, 14.2%) |
86,295 (18.8%) |
859 (26.5%) |
10.0 (9.2, 10.7) |
| Female >55 years (n = 1412, 9.1%) |
47,627 (10.4%) |
417 (12.9%) |
8.8 (7.8, 9.7) |
|
Missing data removed.
CI, confidence interval; GP, general practitioner; HMB, heavy menstrual bleeding
|
Management of HMB (April 2008–March 2016)
Management actions were investigated for the period between April 2008 and March 2016 (221,738 encounters with female patients aged 12–54 years) from 7823 GPs. Among these, HMB was managed 1698 times (7.7/1000 encounters). Pathology testing was the most common management action (85.5/100 HMB problems), with full blood examination (FBE) being the most frequently ordered investigation, followed by ferritin, thyroid function and then hormone assay (Appendix 2). There were 25.7 imaging tests ordered per 100 HMB problems managed, nearly all of which were pelvic ultrasonography (24.4/100 HMB problems).
Medications were also frequently used, with 52.7 medications recorded per 100 HMB problems managed. The combined oral contraceptive pill (COCP) was the most commonly recorded medication (14.7/100 HMB problems), followed by tranexamic acid (7.6/100 HMB problems), norethisterone (7.1/100 HMB problems) and nonsteroidal anti-inflammatory drugs (NSAIDs; 6.9/100 HMB problems). Levonorgestrel/ethinyloestradiol was the most frequently recorded COCP, accounting for approximately two-thirds of all COCP recorded. Mefenamic acid accounted for the majority of all NSAIDs recorded. Medroxyprogesterone and iron therapy were both used at a similar rate (5.4/100 HMB problems).
Patients were referred 21.6 times per 100 HMB problems, the vast majority to obstetricians/gynaecologists (19.7/100 HMB problems). Clinical treatments were provided at a rate of 21.1 per 100 HMB problems, most being counselling, advice and or education (18.8/100 HMB problems). Procedural treatments were rarely performed (3.4/100 HMB problems).
Management of HMB by patient age (April 2008–March 2016)
There were no significant differences in the pathology test order rate by age, but women aged 35–44 and 45–54 years were more likely than female patients aged 12–24 years to have a hormone assay test ordered. Pregnancy tests (urine and/or blood) were more frequently ordered/performed for women aged 25–34 than among those aged 45–54 years. Pap smears were performed significantly more often among patients aged 35–44 and 45–54 years than those aged 12–24 years.
Patients aged 12–24 years were significantly more likely to have HMB managed with a medication, primarily due to a significantly higher prescription rate for a COCP than older age groups. Patients aged 45–54 years had significantly lower rates of COCP compared with all younger age groups. In contrast, the use of a levonorgestrel intrauterine system (LNG-IUS) increased significantly with age, from none recorded for patients aged 12–24 years to 5.8 recorded per 100 HMB problems in women aged 45–54 years.
Patients aged 12–24 years were significantly less likely to have HMB managed with an imaging test (mainly pelvic ultrasonography) or with a referral (mostly to obstetricians/gynaecologists) than women in the older age groups.
Management of new and previously managed (old) HMB problems (April 2008–March 2016)
Problem status (new/old) was specified for 1372 HMB problems (80.8%). New cases numbered 715 (52.1%; 3.2/1000 encounters) and old cases 657 (47.9%; 3.0/1000 encounters).
The rate of medications, clinical treatments or procedural treatments did not differ between new and old HMB problems. However, new cases of HMB generated significantly higher rates of pathology tests (including FBEs, ferritin, thyroid function and hormone assays), imaging orders (pelvic ultrasonography, in particular), Pap smears and pregnancy tests compared with follow-up consultations for HMB. Conversely, patients with an old HMB problem were referred (mostly to obstetricians/gynaecologist) at almost twice the rate of those with a new HMB problem (Table 2).
| Table 2. Management actions per 100 new and old cases of HMB problems managed (April 2008–March 2016) |
| |
Rate per 100 new HMB problems,
(n = 715)
n (95% CI) |
Rate per 100 old HMB problems,
(n = 657)
n (95% CI) |
| Non-pharmaceutical management |
|
|
| Clinical treatments |
24.6 (21.2, 28.1) |
18.9 (15.5, 22.3) |
| Counselling/advice/education |
21.8 (18.6, 25.1) |
16.9 (13.7, 20.1) |
| Procedural treatments |
2.9 (1.6, 4.3) |
3.8 (2.2, 5.4) |
| Local injection/infiltration |
0.3 (–) |
1.5 (0.6, 2.5) |
| Contraceptive device insertion/supply/removal |
1.4 (0.5, 2.3) |
0.9 (0.2, 1.6) |
| Pathology tests |
109.4 (97.0, 121.8) |
62.4 (52.2, 72.6) |
| Full blood count |
27.1 (23.8, 30.5) |
18.1 (15.2, 21.1) |
| Ferritin |
24.3 (21.2, 27.5) |
15.8 (13.0, 18.7) |
| Thyroid function |
13.8 (11.3, 16.4) |
5.6 (3.9, 7.4) |
| Hormone assay |
12.7 (9.4, 16.0) |
3.7 (1.9, 5.4) |
| Coagulation studies |
1.7 (0.7, 2.6) |
1.5 (0.6, 2.5) |
| Imaging tests |
36.5 (32.8, 40.2) |
14.3 (11.5, 17.1) |
| Pelvic ultrasonography |
35.1 (31.5, 38.7) |
12.9 (10.4, 15.5) |
| Referrals |
14.4 (11.8, 17.0) |
29.8 (26.0, 33.6) |
| Obstetrician/gynaecologist |
14.0 (11.4, 16.5) |
27.9 (24.2, 31.5) |
| Pharmaceutical management* |
53.6 (49.0, 58.2) |
53.0 (48.2, 57.7) |
| Combined oral contraceptive pill† |
16.4 (13.6, 19.1) |
13.9 (11.2, 16.5) |
| Levonorgestrel/ethinyloestradiol |
11.6 (9.3, 14.0) |
7.9 (5.8, 10.0) |
| Cyproterone/ethinyloestradiol |
1.7 (0.7, 2.6) |
0.6 (0, 1.2) |
| Drospirenone/ethinyloestradiol |
1.4 (0.5, 2.3) |
2.1 (1.0, 3.2) |
| Norethisterone/ethinyloestradiol |
1.3 (0.4, 2.1) |
2.0 (0.9, 3.0) |
| Tranexamic acid |
6.4 (4.6, 8.3) |
8.2 (6.6, 11.0) |
| Norethisterone |
7.8 (5.8, 9.8) |
6.4 (4.4, 8.3) |
| Medroxyprogesterone† |
4.3 (2.3, 6.4) |
6.1 (3.8, 8.4) |
| Depot medroxyprogesterone acetate† |
0.4 (0, 0.9) |
2.4 (1.3, 3.6) |
| Levonorgestrel intrauterine system† |
3.2 (1.9, 4.6) |
5.6 (3.8, 7.4) |
| Nonsteroidal anti-inflammatory drugs† |
8.3 (6.2, 10.3) |
6.5 (4.6, 8.5) |
| Mefenamic acid |
5.9 (4.1, 7.6) |
5.2 (3.4, 6.9) |
| Iron therapy† |
5.5 (3.8, 7.1) |
4.7 (3.1, 6.3) |
| |
Rate per 100 new HMB encounters |
Rate per 100 old HMB encounters |
| Pap smears‡ |
13.7 (11.1, 16.3) |
7.0 (5.0, 9.0) |
| Pregnancy test‡ |
3.6 (2.3, 5.0) |
0.6 (0.01, 1.2) |
*Includes all medications prescribed, supplied or recommended for over-the-counter purchase
†Classification according to the Coding Atlas of Pharmaceutical Substances and the Anatomical Therapeutic Classification System outlined in Appendix 1
‡Includes all pregnancy tests and Pap smears performed or ordered
CI, confidence interval; HMB, heavy menstrual bleeding |
Discussion
This study revealed an increased number of GP–patient encounters for HMB in Australian general practice from 2000 to 2016. This rise may be due to increased awareness among women seeking care for HMB, increased opportunistic diagnosis and management by GPs or a combination of both.
Management rates during this period were highest among women aged 35–44 years, followed by those aged 45–54 years. The higher management rates in mid- and older-reproductive age women are consistent with the presentation of HMB associated with perimenopausal hormonal fluctuations, and the increased risk of malignancy or other serious pathology in women with increasing age. The lower rates of management among younger women may be attributable to less awareness of what constitutes normal menstrual blood loss, a reluctance by patients to raise the issue, especially if seen by a male GP, or not being opportunistically asked about menstrual bleeding patterns during a routine cervical screening consultation. The use of hormonal contraception may also subsequently reduce the likelihood of HMB. The more frequent management of these problems by female GPs is likely to reflect the fact that a very large proportion of encounters with female GPs are with female patients, and female GPs manage female genital problems significantly more often than their male counterparts.10
GP management of HMB in 2008–16 was found, appropriately, to vary between patient age groups, as management is dependent on the severity and likely cause of bleeding, future fertility plans, medical contraindications and personal preferences. The six quality statements from the HMB clinical care standard that are relevant to general practice highlight some areas for quality improvement (Box 1).3
Statement 1 advises routinely excluding pregnancy, iron deficiency and anaemia at the initial assessment. Pregnancy can be generally excluded by a clinical history, and pregnancy tests were rarely ordered/performed at HMB encounters, including for new cases. While FBE and serum ferritin were the most commonly ordered pathology tests for the management of new HMB problems, testing rates were relatively low.
Statement 2 recommends supporting informed choice and shared decision making. The rates of counselling, advice and education were relatively low, even among new cases, and might reflect inconsistent documentation. Further studies are required to evaluate the quality of the information provided.
Statement 3 recommends women be offered pharmaceutical treatment before uterine-preserving procedural options, such as endometrial ablation or surgical management with hysterectomy, are considered. If hormonal and non-hormonal pharmaceutical treatments are acceptable and malignancy is excluded, the LNG-IUS should be considered, followed by tranexamic acid, NSAIDs or COCPs. Cyclic high-dose norethisterone or injectable long-acting progestogens (depot medroxyprogesterone) can also be considered, but their use may be limited by side effects.3 Women undergoing further investigations should also be offered immediate symptomatic treatment to reduce blood loss in subsequent menstrual cycles.
While the COCP was prescribed relatively frequently for both new and old HMB, the rate of prescription of iron therapy was low, which may suggest suboptimal management for immediate symptom relief for some women. Tranexamic acid and NSAIDs were less frequently prescribed than COCP and could be considered more frequently for symptomatic management. Statement 4 recommends pelvic ultrasonography, preferably transvaginal, if structural/histological causes are suspected. Rates of pelvic ultrasonography orders were highest among women aged 45–54 years for new HMB problems. possibly reflecting GP awareness of the need to exclude malignancy and other serious pathology in this older age group.
Statement 5 highlights the need, if clinically appropriate, for women to be offered the LNG-IUS, as it is the most effective medical option for managing HMB.
The BEACH data only record LNG-IUS prescriptions/insertions by GPs and do not reflect prescriptions/insertions by obstetrician/gynaecologists for referred patients. Insertion of an LNG-IUS in a specialist setting after further assessment constitutes appropriate management, especially where serious pathology is suspected, and may partly explain the low prescription rates for the LNG-IUS. However, insertion of an LNG-IUS for HMB after exclusion of serious causes and with appropriate follow up can occur in the GP setting. While not all GPs will insert the LNG-IUS, it is essential that rapid referral pathways for insertion are available to ensure timely access to this management option.
Statement 6 recommends referral to a specialist when there is suspicion of malignancy, other significant pathology or a woman has not responded to medical treatment after six months. The higher rates of specialist referrals seen at old problem status encounters is not surprising, as referral is more likely after investigations have been undertaken and may reflect appropriate care being provided to women most at risk of serious pathology.
The strength of this study is its large representative sample of GP management of HMB. However, many of these standards require context that may be difficult to fully measure using cross-sectional data. BEACH data do not measure care provided outside the encounter studied or the GPs’ existing knowledge of the patient.
In conclusion, this study provides insights into the management of HMB in Australian general practice in a period prior to the release of the HMB clinical care standard and serves as a benchmark for future research into the impact of the clinical care standard on future care provided. It highlights areas for quality improvement to ensure the right patients receive the right care at the right time. The results also support the need for continued research, education and training to address the barriers and enablers for the use of the LNG-IUS and to facilitate uptake in line with the recommendations of the HMB clinical care standard.