Case
A man aged 55 years presented with a two-week history of an enlarging painful ulcer affecting his right lower leg. It had not responded to a five-day course of oral cephalexin. He had no prior history of skin ulcers. His medical history included hypertension, gout, obesity and chronic obstructive pulmonary disease. On systems review, the patient stated he had not experienced any fevers, sweats, arthralgia, rashes, oral ulcers, diarrhoea, haematochezia, melaena or weight loss.
On examination, he was afebrile and haemodynamically stable. His heart sounds were dual with no murmurs. Above his right medial ankle was an ulcer with a raised, violaceous edge with surrounding erythema (Figure 1). The beginnings of multiple new smaller ulcers were also noted to be present on both lower legs.
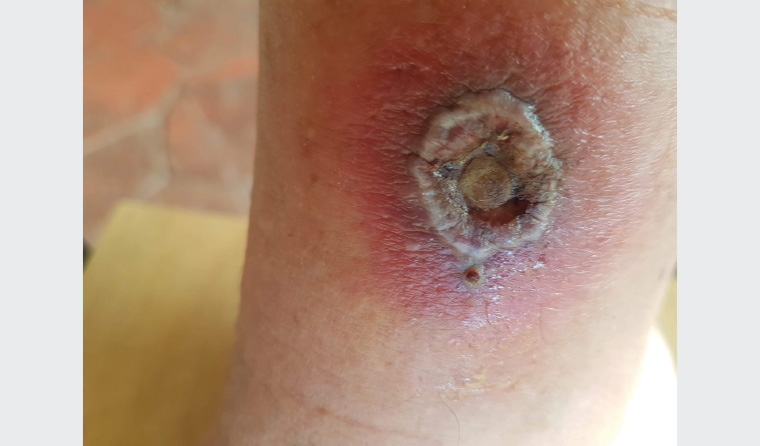
Figure 1. Photograph of the ulcer
Question 1
What are the possible causes of this ulcer?
Answer 1
Causes of skin ulceration include exogenous injury, venous insufficiency, neuropathy, infections, arterial insufficiency, cutaneous cancers, systemic vasculitis, manifestations of autoimmune and connective tissue disease, and neutrophilic dermatoses such as pyoderma gangrenosum.
Case continued
Three 4 mm punch biopsies were taken from the ulcer edge for histopathology, immunofluorescence and tissue microscopy, culture and sensitivities (MCS). A swab of the ulcer for MCS was also taken. No pathogens were isolated from these cultures. Specific tests for mycobacterial and fungal infections were not requested. Histopathology showed a prominent neutrophilic infiltrate (Figure 2), and there were no organisms seen with an alcian blue/periodic acid–Schiff stain. The superficial dermal vessels showed features of vasculitis. Immunofluorescence was negative for immunoglobulin (Ig) G, IgA, IgM, fibrinogen and C3.
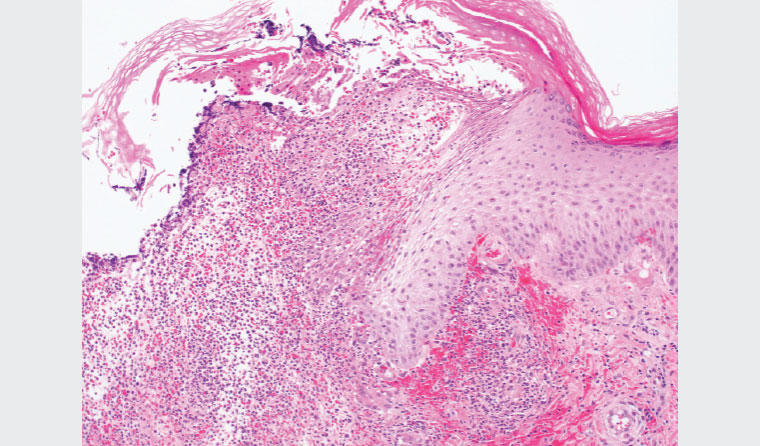
Figure 2. Micrograph involving the ulcer edge showing necrosis and a prominent inflammatory infiltrate of neutrophils
There was no leucocytosis and no neutrophilia on the full blood examination. The C-reactive protein was 5 mg/L (reference range <5 mg/L). Pathology tests to screen for inflammatory bowel disease, inflammatory arthritis and haematological disorders were negative. A provisional diagnosis of pyoderma gangrenosum was discussed with the patient.
Question 2
How is pyoderma gangrenosum diagnosed?
Question 3
What conditions are associated with pyoderma gangrenosum, and what is the epidemiology?
Question 4
What are the treatment options for pyoderma gangrenosum?
Answer 2
Pyoderma gangrenosum is a clinical diagnosis and a diagnosis of exclusion. Misdiagnosis and delayed diagnosis are common. The key investigation is an adequate deep incisional biopsy of an ulcer edge, accepting risk of pathergy. The Mayo Clinic proposed diagnostic criteria in 2004 (Table 1).1
New diagnostic criteria have recently been proposed in order to eliminate the diagnosis of exclusion criterion.2
| Table 1. Mayo Clinic proposed diagnostic criteria for ulcerative pyoderma gangrenosum1 |
| Major criteria (both required) |
Minor criteria (two or more required) |
- Rapid progression of a painful necrolytic cutaneous ulcer with an irregular, violaceous and undermined border
- Exclusion of other causes of cutaneous ulceration
|
- History suggestive of pathergy or clinical finding of cribriform scarring
- An associated systemic disease
- Histopathological findings (sterile dermal neutrophilia +/– mixed inflammation +/– lymphocytic vasculitis)
- Treatment response
|
Answer 3
An associated systemic condition is present in about half of patients. Inflammatory bowel disease, inflammatory arthritis and haematological disorders are the most common.3 Screening for associations is often part of the evaluation of pyoderma gangrenosum. A number of medications can induce pyoderma gangrenosum, with their withdrawal typically leading to remission.4 There are limited epidemiological data available because of the rarity of pyoderma gangrenosum. It is estimated to affect 3–10 people per million population per year.5 It can occur in all age groups but most commonly occurs in those aged 40–59 years and has a slight female predominance.6
Answer 4
A variety of immunosuppressive medications have shown efficacy in the treatment of pyoderma gangrenosum but, because of the lack of evidence-based guidelines, the ideal timing, order and combination of use is not known.7 A stepwise approach is advocated, beginning with optimal wound care, prevention of pathergy, adequate analgesia and a potent topical corticosteroid (intralesional administration has been reported). Patient healing factors should be addressed; for example, smoking cessation, glycaemic control and treating oedema from venous insufficiency. The next step is the addition of systemic therapy, with prednisolone considered first line dosed at 0.5–1 mg/kg/day.8 Typically a response is noted within 2–3 days. Cyclosporine is a suitable alternative in cases resistant to adequately dosed prednisolone. Approximately 50% of patients will achieve remission following six months of treatment with prednisolone or cyclosporine.9 Adjunctive medications such as methotrexate can be used to reduce the requirement for corticosteroids. Biological agents such as infliximab are effective10 but in Australia are not approved by the Therapeutic Goods Administration or listed on the Pharmaceutical Benefits Scheme for the treatment of pyoderma gangrenosum, thus requiring case-by-case consideration.
Case continued
The patient was referred to dermatology and commenced on oral prednisolone. The dose needed to be increased to 75 mg daily to achieve a response. Methotrexate was commenced to a target dose of 25 mg weekly to minimise long-term use of prednisolone. The ulcers were managed with wound care and an ultrapotent topical corticosteroid, later changed to topical tacrolimus. Eighteen months later, the patient continued to take prednisolone and methotrexate. A condition associated with his pyoderma gangrenosum was not identified.
Key points
- It is important to consider pyoderma gangrenosum when evaluating painful, rapidly progressing ulcers, especially on the lower limbs.
- The key investigation is a deep incisional biopsy of an ulcer edge.