Obesity is a chronic condition prevalent in Australia, affecting one-third of the population.1 Defined by the World Health Organization (WHO) as a body mass index (BMI) >30 kg/m2,2 obesity presents significant health risks and often requires medical interventions when lifestyle changes fail. Bariatric surgery is the most effective treatment for obesity, with proven long-term durability in studies with 20 years’ follow-up. The 2022 national health survey showed rising obesity in Australia over the last 15 years, with weight data recorded for 19.71 million Australians aged ≥18 years. In total, 7.9% of Australian adults have a BMI between 35 and <40 kg/m2, with 4.6% having a BMI of ≥40 kg/m2. All of these adults qualify for bariatric surgery. An additional 3.75 million patients with a BMI >30 kg/m2 might qualify, based on other health parameters.1
This paper aims to summarise the procedures, efficacy, indications, contraindications, postoperative care and long-term management, focusing on what Australian general practitioners (GPs) need to know when considering referral and their own practice.
Australian registry data for 2023 show 15,985 primary bariatric operations performed. Based on numbers from the latest national health survey, this would equate to only 0.65% of the eligible population who have a BMI >35 kg/m2.1 Sleeve gastrectomy (SG) made up 80% of cases, 11% comprised one-anastomosis gastric bypass (OAGB), 9% comprised Roux-en-Y gastric bypass (RYGB) with any other type of procedure accounting for under 1%.3 Bariatric surgery is safe, with deaths from any cause within 90 days of surgery at 0.04% for primary and 0.05% for revisional operations in Australia.4 Adverse events within 90 days of surgery including unplanned return to theatre, unplanned return to an intensive care unit or hospital readmission, was 1.5% following SG, 3.8% following OAGB and 4.8% following RYGB in 2023. Only 5% of cases in Australia were performed in the public healthcare system.3
Indications
Indications for surgery were updated in 2022 by a joint statement from the American Society of Metabolic and Bariatric Surgery (ASMBS) and the International Federation for the Surgery of Obesity and Metabolic Disorders (IFSO).4 Bariatric surgery is recommended for patients with a BMI >35 kg/m2, regardless of obesity-related comorbidities. It is also advised for individuals with a BMI >30 kg/m2 who have type 2 diabetes. Surgery can also be considered for patients with a BMI <35 kg/m2 if non-surgical methods have not led to significant weight loss or improvement in obesity-related comorbidities. Other indications include a bridge to other treatments such as joint arthroplasty, abdominal wall hernia repair and organ transplantation.5
Contraindications
Bariatric surgery is generally safe for most patients, with no strict contraindications outlined in the recent guidelines.5 However, certain conditions require careful evaluation and management, including frailty, paediatrics and adolescents, cirrhosis, heart failure and mental health issues. Disordered eating, severe uncontrolled mental illness and active substance abuse are particularly critical considerations.5
Efficacy
Surgery is the most effective long-term treatment for obesity, with maintenance of weight loss that is sustained for decades. The Swedish obese subjects (SOS) trial provides data on long-term weight loss and resolution of comorbidities; there was an 18% total weight loss (TWL) following surgery compared to –1% in controls at 20 years.6 Two randomised controlled trials (RCTs) have compared SG and RYGB. SLEEVEPASS and SM-BOSS trials both showed excellent long-term weight loss with similar outcomes for each procedure.7,8 At 10 years, SG resulted in a TWL of 23.4% compared to 26.9% for RYGB.7
Bariatric surgery typically results in approximately 30% TWL initially, which is then followed with some weight regain, before patients achieve a long-term weight plateau. This regain of weight is partly due to improved dietary tolerance and a decrease in metabolic rate. It is important to reassure patients that this pattern of some weight regain is normal and not a failure, as they ultimately maintain substantial weight loss. Combined RCT data show TWL after both SG and RYGB at 1 year to be 28.2% versus 30.8%, at 3 years to be 26.5% versus 29.9%, and at 5 years to be 23.7% versus 27.2%, respectively (Figure 1).9
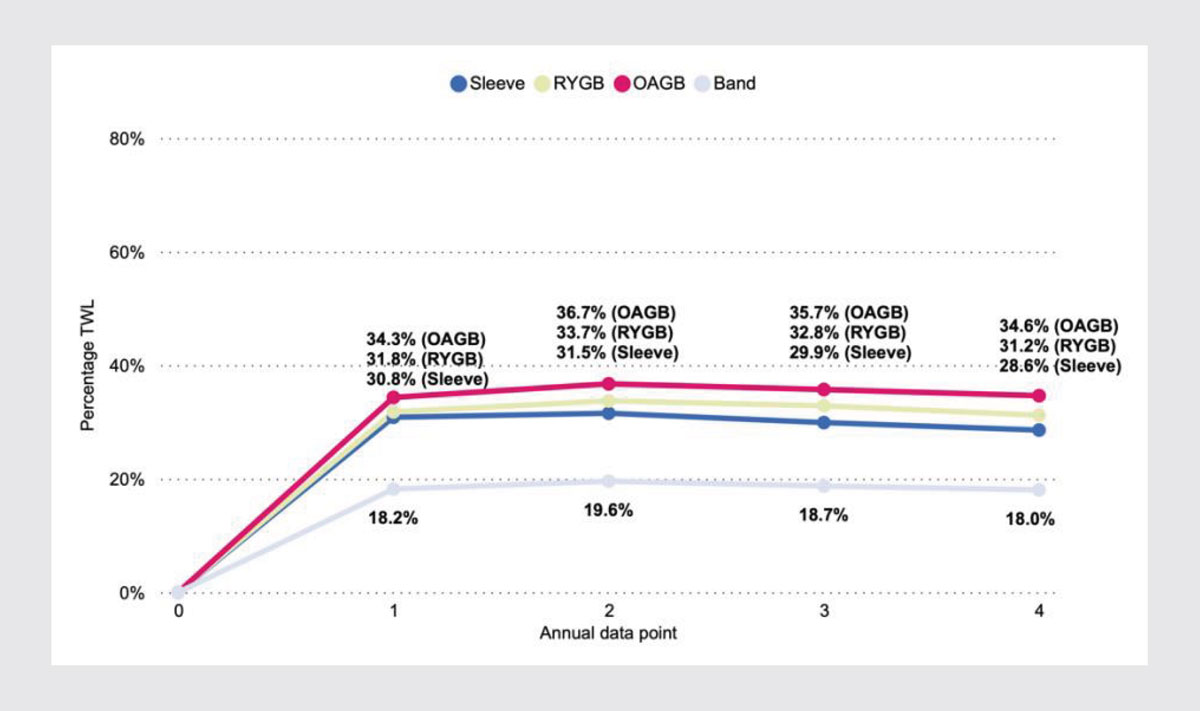
Figure 1. Average percentage TWL for adult primary participants who have annual weight data for the first four years by procedure type in Australia (N=6229).2
Includes participants who have had a conversion procedure and excludes participants who do not have weight data for all of the first four annual data collection points and those with other types of primary procedures.
Band, laparoscopic adjustable gastric band (n=1902); OAGB, one-anastomosis gastric bypass (n=254); RYGB, Roux-en-Y gastric bypass (n=538); Sleeve, laparoscopic sleeve gastrectomy (n=3535); TWL, total weight loss.
Reproduced from The Australian and New Zealand Bariatric Surgery Registry. Bariatric surgery registry. 2023 annual report. Australia Bariatric Surgery Registry, 2023. Available at www.monash.edu, with permission from The Australian and New Zealand Bariatric Surgery Registry.3
In comparison, diet and lifestyle changes typically result in a 5–10% TWL. Glucagon-like peptide-1 (GLP-1) agonists offer better outcomes, with liraglutide achieving an 8.0% TWL at 56 weeks and semaglutide a 14.9–16.0% TWL at 68 weeks. Treatment with tirzepatide, a GLP-1/gastric inhibitory polypeptide (GIP) agonist, led to a 20.9% TWL at 72 weeks when 15 mg was taken weekly. Treatment needs to be continued for maintenance of weight loss. This evolving landscape emphasises the need for combined treatment approaches, including medication and surgery. Endoscopic procedures such as the intragastric balloon initially showed a 10.2% TWL at six months, but this decreased to 7.6% six months after balloon removal. Endoscopic sleeve gastroplasty resulted in a 13.6% TWL at 12 months, though long-term data on these procedures are still limited.10
Comorbidity resolution
The effect on diabetes control has been extensively investigated following bariatric surgery. The STAMPEDE trial compared RYGB and SG against best medical therapy alone. The primary outcome of a haemoglobin A1c (HbA1c) level of 6.0% or less was achieved in 29% who underwent RYGB and 23% who underwent SG, compared with 5% who received medical therapy alone. The mean percentage of HbA1c was reduced by 2.1% in the surgery group compared to 0.3% who received medical therapy alone. Insulin use reduced by 35%, 34% and 13% in the RYGB, SG and medical therapy groups, respectively. TWL at five years was 23%, 19% and 5%, respectively. A significantly lower number of patients were taking glucose-lowering medications following RYGB (45%) compared with SG (25%).
11
Surgery has improvements for many other comorbidities. Overall mortality risk is reduced by almost 30% (hazard ratio [HR] 0.71, 95% confidence interval [CI] 0.54–0.92,
P=0.01).
6 Cardiovascular deaths reduced following surgery (HR 0.47, 95% CI 0.29–0.76,
P=0.002) as did total first time cardiovascular events of myocardial infarction or stroke (HR 0.67, 95% CI 0.54–0.83;
P<0.001).
12 Heart failure also responds favourably to bariatric surgery.
5
Bariatric surgery leads to improvement in or resolution of obstructive sleep apnoea, asthma, joint disease, reflux, metabolic-associated fatty liver disease (MAFLD), renal dysfunction and urinary incontinence in patients with obesity. It reduces risk of gastrointestinal, genito-urinary, reproductive and haemopoietic malignancies. Quality of life improves proportionate to the amount of weight lost. Weight loss improves participation in physical exercise programs, reduces sick leave and pension payments for disability.
13
Procedures
Vertical banded gastroplasty (VBG) was introduced in 1982 but was phased out due to long-term complications. Patients still present with issues related to outlet obstruction. The adjustable gastric band (AGB), developed in 1986, was once the most common weight loss procedure performed in Australia. Due to modest outcomes and complications, many have been reversed or converted (most commonly to RYGB). AGB comprised only 0.3% of bariatric procedures performed in Australia in 2023. Common complications include reflux, pouch dilation, slippage and food bolus obstruction. Band erosion and port-related issues can also occur.
3
The RYGB, first described in 1967, remains popular for its long-term outcomes and manageable complications. SG, adopted as a stand-alone operation approximately 20 years ago, has become the most common procedure in Australia and worldwide due to its effectiveness and favourable complication rates.
14 Recently, the OAGB, a variant of the RYGB, has risen in popularity and accounts for just over half of gastric bypass procedures in Australia. It demonstrates slightly higher efficacy and lower complication rates compared with RYGB.
3
Sleeve gastrectomy
The laparoscopic SG procedure removes the greater curve and fundus of the stomach, leaving a narrow tube-like structure, which restricts food intake while preserving the pylorus. Benefits include an operation restricted only to the stomach and maintaining endoscopic access to the duodenum. Short-term risks include staple line leak and bleeding. Long-term risks mostly relate to increased reflux with proton pump inhibitor (PPI) intake; 64% at 10 years compared with 12% preoperatively.
7 This creates a theoretical increased risk of Barrett’s oesophagus, a premalignant condition that predisposes to oesophageal cancer; however, Barrett’s oesophagus was measured at 4% in the SLEEVEPASS trial, which was no higher than in the RYGB group.
7 Regular gastroscopy is recommended for monitoring reflux-related oesophageal complications while long-term data are obtained in this area (Figure 2).
15

Figure 2. Sleeve gastrectomy.
Reproduced from Levent E, courtesy of the International Federation for the Surgery of Obesity and Metabolic Disorders (IFSO). IFSO atlas of bariatric and metabolic surgery. IFSO, 2018. Available at www.ifso.com, with permission from Levent E.
Roux en Y gastric bypass
The RYGB involves the creation of a small gastric pouch and connecting it to the jejunum, bypassing part of the stomach and small intestine. Food entering immediately into the mid jejunum promotes satiety and improves glycaemic control through gut hormone release (eg GLP-1, GIP, PYY). The RYGB is highly effective in controlling diabetes and reflux and is ideal for conversion from an AGB. Short-term risks include anastomotic leak and stenosis, long-term risks include anastomotic (marginal) ulcer (especially in smokers) and internal hernias. Long-term dietary management is required to avoid dumping syndrome (Figure 3).

Figure 3. Roux en Y gastric bypass.
Reproduced from Levent E, courtesy of the International Federation for the Surgery of Obesity and Metabolic Disorders (IFSO). IFSO atlas of bariatric and metabolic surgery. IFSO, 2018. Available at www.ifso.com, with permission from Levent E.
One-anastomosis gastric bypass
The OAGB is similar to the RYGB, but with a longer gastric pouch and increased length of bypassed jejunum. It offers a less complex operation, greater weight loss and comorbidity resolution with lower overall risk than RYGB. Complications are comparable, though patients are still prone to acid and bile reflux, whereas internal hernia is rare (Figure 4).
3

Figure 4. One-anastomosis gastric bypass.
Reproduced from Levent E, courtesy of the International Federation for the Surgery of Obesity and Metabolic Disorders (IFSO). IFSO atlas of bariatric and metabolic surgery. IFSO, 2018. Available at www.ifso.com, with permission from Levent E.
Postoperative care
Patients usually have a median two-night postoperative inpatient admission following surgery.
4 Patients progress through a prescribed diet, starting with fluids only, then progress through puree, soft and regular food intake, as per their surgeon’s discretion. In our practice, patients are advised to have liquids for two weeks, then puree for two weeks before soft food for two weeks. Initially, a patient’s oral intake is quite restricted, and the focus is on maintenance of hydration and protein intake. Patients often need advice regarding the dietary progression and management of pain and nausea. Regular PPI treatment is recommended during the early postoperative phase. Daily multivitamin intake is important due to the frequency of micronutrient deficiency.
Constipation is also a common issue because of the decreased fibre intake postoperatively as well as decreased fluid intake. Patients are also often on opiate medications during the initial few days. Patients should be encouraged to walk short distances frequently in the first week or two, and then progress to regular exercise as they are able to maintain better oral intake.
Pre-existing medical comorbidities and their medication requirements can change rapidly after surgery. A review every 1–2 weeks initially for adjustment might be required, particularly for patients who are taking diabetes and antihypertensive medications.
Long-term management
Weight loss is substantial in the first few months after bariatric surgery. Long-term care focuses on managing lifestyle changes, with regular multivitamin use and annual nutritional screening tests essential to prevent common deficiencies.
From a study reporting long-term deficiencies after RYGB or SG, vitamin and mineral deficiencies with over 10% prevalence included: Hb, iron and vitamins B1, B3, B6, B9, B12, A, C, D, E, along with parathyroid hormone (PTH), magnesium, zinc and selenium.
16
Conversion surgery rates are under 5% at 5 years for SG and RYGB, whereas AGB, now rarely performed, has over 15% conversion rates.
3 Revision or conversion surgery is considered for significant weight regain or complications such as reflux, with medical therapy explored as the first option.
Patients who have undergone SG or OAGB should maintain regular long-term reflux surveillance, particularly for Barrett’s oesophagus, with checks at one year and every 2–3 years thereafter advised. Following AGB or RYGB, gastroscopy is only needed for investigation of symptoms, as for the general population.
15 Ongoing endoscopic surveillance for pre-existing indications such as Barrett’s oesophagus should be continued.
Long-term, patients reach their nadir weight within 1–2 years. Some regain is typical before stabilising around five years, with most maintaining this weight long term. Understanding this pattern helps set realistic expectations for patients.
Conclusion
Bariatric surgery is the most effective long-term treatment for obesity. Although other treatments show promise, surgery remains the best option for eligible patients willing to pursue it. Surgery is safe, with mortality at 0.04% for primary procedures. SG accounts for 80% of primary bariatric procedures, whereas gastric bypasses make up the remaining cases (11% OAGB and 9% RYGB). Initial TWL averages around 30%, with most maintaining significant TWL (approximately 25%) at 10 years. Overall complication rates within 90 days are 1.5% for SG, 3.8% for OAGB and 4.8% for RYGB. Long-term management includes lifestyle counselling, adherence to multivitamin supplementation, annual nutritional screening and endoscopic surveillance after SG and OAGB.
Key points
- Bariatric surgery has the most effective long-term weight loss result and comorbidity resolution compared to other treatments, with low complication (1.5–4.8%) and mortality rates (0.04–0.05%).
- Surgery typically results in 30% TWL initially, stabilising to around 25% long term.
- Bariatric surgery is recommended for patients with a BMI >35 kg/m2 or a BMI >30 kg/m2 and type 2 diabetes.
- Early postoperative care involves diet progression, management of pain, nausea and constipation, PPI and multivitamin use.
- Long-term management involves lifestyle counselling, multivitamin use, annual nutritional screening and regular endoscopic surveillance following SG and OAGB.