Motor neurone disease (MND) encompasses a group of neurodegenerative disorders including amyotrophic lateral sclerosis (ALS; the most common form of MND), primary lateral sclerosis, progressive muscular atrophy and progressive bulbar palsy.1 Approximately 90% of ALS cases are sporadic. The remaining 10% of cases are familial, involving mutations in genes such as the superoxide dismutase type 1 gene and C9ORF72 gene.2 A family pedigree, genetic counselling and the involvement of a geneticist are useful in familial ALS.3
Protein aggregation, glutamate, neuronal excitotoxicity and oxidative stress have been implicated in the development of ALS.4,5 However, the exact aetiology of ALS is yet to be clearly elucidated.
Diagnosis is made on the basis of clinical findings, supplemented by targeted investigations, including electromyography and nerve conduction studies. MND is a diagnosis of exclusion. A hallmark of ALS is the presence of both upper and lower motor neurone signs, including limb weakness, hyperreflexia and fasciculations. Frontotemporal dementia may be present.6 Evidence of denervation and re-innervation, involving a number of muscles, may be seen on electromyographic studies.7
Patients may present to the general practitioner (GP) with asymmetric limb weakness, dysphonia or dysphagia. The diagnosis is often delayed, as patients may be presumed to have an isolated disorder of the peripheral nervous system, for example ‘foot drop’ secondary to compressive lumbar neurospinous disease, or a laryngeal condition. This may result in an initial referral to a range of specialists, such as a neurosurgeon or ear, nose and throat (ENT) surgeon, before eventual referral to a neurologist. As a result, the mean age at which MND is diagnosed is approximately 64 years, two years after the mean age of symptom onset at 62 years.8
Potential differential diagnoses include multifocal motor neuropathy, cervical myelopathy, chronic inflammatory demyelinating polyneuropathy, Kennedy’s disease and post-polio syndrome.
Given its progressive and debilitating nature and the lack of cure, management of MND is focused on symptomatic relief, optimising quality of life and timely palliative care involvement. Early referral to a dedicated MND multidisciplinary clinic is important to address the multiple facets and highly specific and complex individual care needs of this group.
MND in Australia
MND affects approximately nine in every 100,000 people in Australia, with marginally more males affected than females and a mean survival of 2.5 years from the time of diagnosis to death.9,10 Approximately 750 deaths each year are due to MND, with respiratory failure being the most common cause of death.9,10 Therefore, it is advantageous for GPs to be well informed regarding the management of MND symptoms, including sialorrhoea, spasticity, pain and respiratory dysfunction. Table 1 outlines common MND issues and management options. Patients often require non-invasive ventilatory support and either a percutaneous endoscopic gastrostomy (PEG) or a radiologically inserted gastrostomy (RIG) tube.11
|
Table 1. Common issues and management options in motor neurone disease
|
|
Common issues
|
Management
|
|
Riluzole use
|
|
|
Sialorrhoea
|
-
Anticholinergic agents (eg combination preparations containing hyoscyamine, atropine and hyoscine), atropine drops orally, glycopyrrolate (nebulised or subcutaneous)
-
TCAs (eg amitriptyline)
-
Injections of botulinum toxin into salivary glands26
-
Low-dose, unilateral, salivary gland radiotherapy27
|
|
Spasticity
|
-
Muscle relaxants (eg baclofen, dantrolene sodium – monitor LFTs closely)
-
Pregabalin, gabapentin, cyproheptadine, clonidine
-
Botulinum toxin injections
|
|
Viscous/tenacious sputum
|
-
Frozen dark grape juice (contains mucolytic enzymes)
-
Nebulised normal saline
-
Mucolytic agents (eg bromhexine, nebulised N-acetyl cysteine)
|
|
Respiratory dysfunction
|
-
Monitor with respiratory function tests, polysomnography
-
Non-invasive ventilatory support (eg BiPAP)
-
Low-dose benzodiazepines, morphine
|
|
Bulbar dysfunction
|
-
Early speech pathology involvement
-
Dysphagia and aspiration: modified diet consistency (eg puree, fluid thickening agents, modified cups and utensils, manoeuvres to limit laryngeal penetration; enteral feeding)
-
Dysphonia/hypophonia/aphonia: voice amplification systems (eg lapel microphone, communication boards, Boogie Board tablet, smartphone and iPad apps, eye gaze technology)
|
|
Pain
|
-
Paracetamol, anti-inflammatory drugs, opioid analgesia, gabapentin, pregabalin, TCAs, intra-articular steroid injections, physiotherapy, TENS
|
|
Constipation
|
-
Maintain adequate hydration
-
Aperients, suppositories, enemata
-
Cessation, where possible, of drying agents (eg anticholinergics, diuretics)
|
|
Weight loss
|
-
Early involvement of a dietitian
-
Treatment of symptoms such as gastro-oesophageal reflux disease and nausea
-
Nutritional supplements
-
Timely insertion of PEG or RIG tubes and transition to enteral feeding
|
|
Mood disorders
|
-
‘Coffee club’ for patients and carers
-
Social work and pastoral care input
-
Anxiolytics and antidepressants
|
|
Xerostomia (uncommon)
|
-
Maintaining adequate hydration
-
Artificial oral saliva preparations
-
Oral pro-cholinergic drops (eg pilocarpine)
-
Cessation, where possible, of drying agents (eg anticholinergic, diuretics)
|
|
Geneticist (where relevant)
|
|
|
Psychiatry and psychology
|
-
Diagnostic (eg frontotemporal dementia)
-
Therapeutic (eg mood disorders, emotional lability [up to 50% of patients] – TCAs, SSRIs, SNRIs)
|
|
Orthotic management
|
|
|
Community and social supports
|
-
Mobility parking scheme
-
Taxi transport subsidy scheme
-
Companion card
-
Massage/acupuncture (eg Calvary Holistic Healing Centre)
-
Occupational therapy driving and home assessments; self-care retraining; equipment and modifications
-
Community palliative care team
|
|
BiPAP, bilevel positive airway pressure; LFT, liver function test; PEG, percutaneous endoscopic gastrostomy; RIG, radiologically inserted gastrostomy; SNRIs, serotonin–norepinephrine reuptake inhibitors; SSRIs, selective serotonin reuptake inhibitors; TCAs, tricyclic antidepressants; TENS, transcutaneous electrical nerve stimulation
|
Disease-modifying agents
Riluzole, an anti-glutamatergic agent, is a disease-modifying, neuroprotective therapy that prolongs survival in ALS by approximately three months.12 Liver function tests (LFTs) must be monitored because of the risk of hepatotoxicity, which may range from mild elevation of serum transaminase to acute liver failure. Guidelines recommend monitoring LFTs every month for the first three months, then every three months for a further nine months, and yearly thereafter.13
Respiratory symptoms
The signs of respiratory dysfunction are subtle initially; over time, more signs manifest. Often the first signs are regularly waking with a headache (due to nocturnal hypoventilation, resulting in hypoxia and hypercapnia) and daytime somnolence, secondary to disordered sleep. Eventually patients report dyspnoea on exertion, then at rest. Early referral to a respiratory physician is important. As patients develop respiratory failure, non-invasive ventilation, including bi-level positive airway pressure (BiPAP), may be used to assist weakened respiratory muscles, thereby improving sleep duration, quality and efficiency, and energy levels.14 Early onset of respiratory failure is indicative of a poor prognosis.15,16 Non-pharmacological measures to minimise dyspnoea involve upright positioning and careful planning of the day (ie spacing out activities that trigger dyspnoea, such as toileting and showering). Pharmacological management includes the use of low-dose benzodiazepines and low-dose morphine.17
Pain
Patients with ALS tend to experience nociceptive pain rather than neuropathic pain.18 Common causes of pain in patients with spinal-onset ALS include muscle cramps secondary to dysfunction of motor units, and shoulder pain either secondary to overuse or from traction on peri-articular structures from the unsupported weight of the arm due to weakness and wasting of the shoulder girdle musculature. Patients should use armrests on chairs and wheelchairs, and may require a sling that supports the forearm, including the elbow.
Pharmaceutical options include paracetamol, anti-inflammatory agents, opioids, gabapentin, pregabalin, tricyclic antidepressants, intra-articular steroid injections and transcutaneous electrical nerve stimulation (TENS). Second-line agents include mexiletine and levetiracetam; however, these require initiation and monitoring by an experienced team.19
MND polyclinics: Focusing on the patient
Multidisciplinary clinics, when compared with traditional models of standardised outpatient care, improve survival in ALS patients.20,21 MND centres across Australia were established to provide a combination of health services at a single point of contact. Similar care models exist in Europe and North America. GPs caring for patients with MND can refer patients to these clinics by contacting the providers directly; however, often the patient’s neurologist will make the referral immediately following diagnosis. A flowchart of the overall multidisciplinary management of MND is shown in Figure 1. Patients may be seen in a multidisciplinary clinic, an inpatient unit or in the community, depending on their requirements and the stage of their disease.
Health-related instruments that measure quality of life, such as the Revised ALS Functional Rating Scale, are simple yet valuable tools used in these multidisciplinary clinics to assess function and predict survival in ALS patients.22–24
MND polyclinics involve a coordinated team, either physically present or via referral, that includes senior nursing staff, rehabilitation physicians, palliative care physicians, neurologists, gastroenterologists, respiratory physicians, speech pathologists, physiotherapists, occupational therapists, social workers, psychiatrists, psychologists and dietitians.
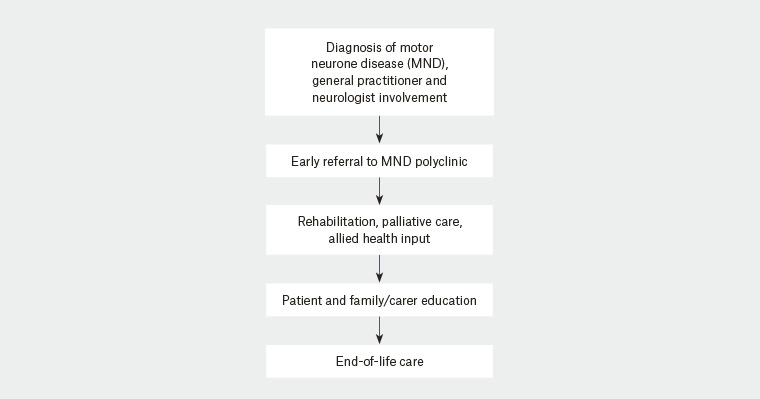
Figure 1. Flowchart of motor neurone disease management and the polyclinic approach
Rehabilitation and allied health
Rehabilitation plays an important part in optimising function and independence, minimising disability and limiting complications. Patients are seen by a rehabilitation physician at their initial clinic visit, during which rehabilitation goals are discussed. Physical therapy, such as strengthening exercises and range-of-motion and mobility re-training, may be beneficial. Physiotherapists also prescribe appropriate walking aids. Patients often report benefit from hydrotherapy.
Occupational therapists provide aids and equipment, which may include hoists, wheelchairs, commodes and hospital beds; these are supplied via Independent Living Centres, Equipment Loan Pools, Enable NSW (or equivalent providers in other states and territories), the Motor Neurone Disease Association and the National Disability Insurance Scheme. Home assessments and driving assessments are conducted, with appropriate modifications for the car, home and workplace.
Patients are reviewed by speech pathologists for the assessment of swallowing and communication. Assistive technology, such as voice amplification, communication boards, smartphone communication apps on tablets or smartphones, and eye gaze technology, are available to patients. Dietitians are involved in the management of nutrition and timely transition to enteral feeding via PEG or RIG tubes.
Community support services, respite and residential care options and end-of-life care planning in terms of wills, enduring power of attorney and enduring guardianship are arranged by social workers within the MND polyclinic. Patients are encouraged to write or record their life history as a means of reflection, as well as leaving a legacy for loved ones. As clinicians, we have observed this to be cathartic for the patient and family prior to, and after, the death of the patient.
Palliative care
The role of palliative care is to offer a holistic approach to the care of the patient by providing adequate symptom relief, respite and planning for end-of-life care and to address psychosocial needs. Early referral to palliative care services allows for symptom management and adequate planning for end-of-life care prior to the severe impairment of speech and cognitive dysfunction. Ideally, the involvement of palliative care should occur from the time of diagnosis, throughout the course of the disease, until the eventual death of the patient and family bereavement.25 Palliative care management is often instituted concurrently with active medical management, and may be in a community, day clinic or inpatient setting.
Common fears of patients faced with imminent death are choking and respiratory distress.25 Having early discussions with the patient and their carers regarding what is to be expected at various phases of the disease, particularly the terminal phase, can allay these fears. Throughout the illness, the role of the carers is pivotal. A significant role for the polyclinic is the physical and emotional support of carers.
End-of-life phase
The end-of-life phase in MND is variable from one patient to another. This phase is marked by any combination of significant functional decline, respiratory failure, dysphagia, marked weight loss, recurrent infection and cognitive decline.25 Symptoms such as dyspnoea can be managed effectively at the end-of-life phase with appropriate pharmacotherapy, such as subcutaneous morphine.
Conclusion
MND is an ‘umbrella’ term for a group of neurodegenerative disorders characterised by an inexorable decline to death. GPs may reasonably expect to encounter at least one patient with MND during their careers, and it is useful for GPs to have a framework to understand the multidisciplinary management of this complex group of diseases.
Resources for general practitioners
- Motor Neurone Disease Australia, www.mndaust.asn.au
- Red flags diagnostic tool for GPs, MND care, www.mndcare.net.au