Chronic diseases are currently the leading cause of ill health and death in Australia.1 Various approaches have been taken to manage these conditions,2 most with limited success. However, these approaches still generally involve a conventional 1:1 model of medical consultation or, alternatively, group education without medical consultation. The need to increase access to services and improve outcomes makes the testing of alternative treatment modalities a priority.
Shared medical appointments (SMAs) are ‘individual medical consultations carried out sequentially in a group of patients with similar concerns, with all listening and contributing’.3 These have been tested extensively in many countries4–8 and have been adopted as a process in the emerging discipline of lifestyle medicine.9
Our work with SMAs in primary care centres6–8 has led us to extend this into a form of ‘programmed’ SMA (pSMA), which potentially provides a platform for a range of interventions for chronic diseases and conditions.
pSMAs have been defined as ‘a sequence of SMAs in a semi-structured form providing discrete educational input relating to a specific topic’.10 These allow for a set number of SMAs run in a sequenced ‘active learning’ format coordinated by an SMA facilitator (practice nurse/allied health professional [AHP]) with generic training in conducting SMAs and specific training in the disease topic, but with a doctor providing individual sequential consultations during part of the session, with participation and input of the group.
The following represents a trial of pSMAs for weight management in primary health care (PHC) centres that include general practitioners (GPs) in NSW.11 Weight management was chosen for testing as obesity is currently a health problem worldwide, and it is a chronic condition for which PHC centres need to become more engaged.12 Preliminary work has also indicated the potential value of such an approach.13 The primary aim of the trial was to measure the acceptability of pSMAs for best use in primary care. Secondary aims were to measure weight loss over a 12-month period and to assess cost and time effectiveness of the process.
Method
Ethics approval for this study was granted by Southern Cross University Ethics Committee (Renewal Approval Numbers ECN-14-268 and ECN-18-181).
Setting
Medical centres
PHC centres in the South Eastern Primary Health Network of NSW were invited to participate through a call for expressions of interest. The aim was completion of six pSMA sessions over 14–16 weeks in each of four pSMA groups (two all-male groups and two all-female groups) with 6–12 participants per group. A selection questionnaire was used to select centres on the basis of our previous work with such centres.
Patients
Overweight and obese (body mass index [BMI] 24–44 kg/m2) patients were recruited through GP referrals and other centre staff on the basis of perceived need and expressed interest. The only exclusions were no English language capability and extreme obesity (BMI >45 kg/m2) with complications. There was no charge for attendance.
Facilitators
Nurses or AHP staff volunteered for training as facilitators for the program. Our goal was to obtain 1–2 facilitators per centre. Staff selection was vetted by the study investigators with PHC staff as having the required qualities for the role of an SMA facilitator as specified by Noffsinger.3
Doctors
Doctors were GPs with no specific background in SMAs or weight control. It was explained that while it may be beneficial for GPs to attend facilitator training, this was not compulsory, as the doctor in an SMA is required to conduct a standard medical consultation (with the possible exception of an intimate examination). It differs from a standard consultation as it is conducted in the presence, and with participation, of other patients. However, doctors were encouraged to familiarise themselves with up-to-date content material relating to weight control on the facilitators’ websites and in a text14 and guidelines provided.15
Procedure
Facilitator training
Selected facilitators underwent two days of face-to-face training. The first day of training focused on conducting SMAs in general (www.lifestylemedicine.org.au/shared-medical-appointments-workshop-for-practitioners-and-facilitators), and the second day focused on evidence-based weight management skills. Successful completion of training resulted in certification by the Australasian Society of Lifestyle Medicine (ASLM).
Program content
The program was designed as an ‘active learning’ initiative based on evidence-based weight loss information. The process of active learning involved all patients participating in the learning process rather than passively listening. It included the optional choice of meal replacements and possibility for triaging into additional psychological care, prescription medication and/or surgical intervention if required.16
Program structure
The program was conducted over six sessions, two weeks apart for the first two sessions, then monthly. The focus was on reducing ‘volume’ of energy input (where volume(i) = energy density × portion size × frequency of eating/drinking) and increasing ‘volume’ of energy expenditure (where volume(e) = intensity × duration × frequency of physical activity). Topics differed for males and females, although there was significant overlap. Each session lasted 90–120 minutes. For the first 20–40 minutes facilitators presented a pre-prepared audiovisual presentation (approximately 10 minutes in length) and held an open discussion on the topic for the session, before the doctor joined for one hour for individual sequential consultations. The facilitator spent 10–15 minutes at the end of the session summarising and answering questions. Participants were given a program handbook, weight loss goals and access to presentations and online links. Each session was designed around three major actions, which were listed on a fridge magnet provided to patients after each session. As in standard SMAs, patients signed a confidentiality agreement and were advised in an introductory brief that confidential matters did not need to be raised, and that a standard 1:1 consultation could be used for this.
Measures
Participant and provider satisfaction were measured using a five-point Likert scale (1 = very negative; 5 = very positive). Weight was taken by facilitators:
- at baseline
- after the 16–18-week intervention at the last SMA
- from medical records from each centre (70%) after 12 months, within a two-month window (ie 11–13 months), or self-reports from follow-up telephone calls when these were not available.
Twelve-month weights were compared with beginning weights for participants who had not completed at least four sessions (‘non-adherers’) and whose weights were available from medical records after 12 months (42 of 54). Waist circumferences were also measured but are not reported here because of inconsistency in measures taken by facilitators and difficulty in assessing changes in women in particular. As well as Likert scales, limited semi-qualitative information was provided from semi-structured interviews with providers. Preliminary cost and time effectiveness estimates were made by comparing weight losses (per kilogram) with those from a comparative 1:1 study.17
Statistical analyses
Regression analyses with generalised estimating equation (GEE) adjustment for repeated observations of the same participants were used to estimate percentage changes in mean weight (kg) over the study period. For each GEE model, a Poisson distribution, log-link function and independent working correlation matrix were used. The dependent variable in each model was weight in kilograms. To conservatively account for loss to follow-up, an additional analysis was performed estimating percentage change in weight from baseline to 12 months by substituting missing weight values with baseline observation carried forward (BOCF). Another analysis calculated the proportions of patients who achieved clinically important weight loss (>5%) from baseline to 12 months, both by using the observed data and more conservatively by assuming those with missing weight values at 12 months did not achieve clinically important weight loss.
Results
Centres, facilitators, doctors and patients
Seven PHC centres in the South Eastern Primary Health Network of NSW were chosen from 15 responders to a call for expressions of interest. A total of 216 overweight and obese patients (83 men and 133 women) aged 35–85 years were recruited for 25 groups (mean 8.7 patients per group). Twelve nurses or AHP staff volunteered for training as facilitators for the program. This included practice nurses (8), a diabetes educator (1), an exercise physiologist (1), a dietitian (1) and a psychologist (1). Seven GPs also participated.
Of 216 patients starting the program, 166 (77%), completed at least four of six sessions (hereafter called ‘adherers’). Weight measurements were obtained at 12 months for 89% (n = 192) of participants (92% [n = 153] of all adherers and 78% [n = 39] of non-adherers).
The mean age of both male and female adherers was 57 years. The mean age of those failing to complete four or more sessions was 59 for males (n = 19) and 48 for females (n = 31). Both adherers and non-adherers claimed to have been ‘overweight’ for a mean of 16 years. Socioeconomic status of most participants was estimated by providers as being predominantly low-to-middle income. Mean patient weight loss expectation for male adherers was 21% of current weight; for female adherers it was 25% of current weight. This was similar for non-adherers at 25%.
Tables 1–3 show patient satisfaction with the program and program components.
The participating doctor (used by all) was rated highly as part of the program, confirming the notion of an SMA (Table 3).
| Table 1. Mean participant satisfaction, preferences and recommendation |
| |
Response |
| |
Men
(n = 65) |
Women
(n = 102) |
Total (n = 167)
|
| Satisfaction |
Mean Likert score |
| How do you rate the program you have attended here? |
4.4 |
4.2 |
4.3 |
| How useful has the program been for you? |
4.3 |
4.2 |
4.2 |
| How did the program compare with other weight loss methods you have tried? |
4.2* |
3.2 |
3.7 |
| Preferences |
Yes/Maybe (%) |
| Would you recommend this form of weight loss program to others? |
51 (78%)* |
90 (88%) |
142 (85%) |
| Would you prefer this type of program for weight control over a 1:1 session with your doctor? |
42 (65%) |
83 (81%) |
122 (73%) |
| *Men had tried far fewer weight loss methods than women. |
| Table 2. Mean program component satisfaction ratings of participants |
| Program component |
Mean Likert score |
| |
Men (n = 65) |
Women (n = 102) |
Total (n = 167) |
| Having time for asking questions |
4.5 |
4.4 |
4.4 |
| Seeing the doctor more relaxed than usual |
4.3 |
4.3 |
4.2 |
| Having the doctor’s/staff’s full attention |
4.3 |
4.5 |
4.3 |
| Getting support from others in the group |
4.2 |
4.3 |
4.1 |
| Contribution of other health professionals |
4.4 |
4.3 |
4.2 |
| Hearing experiences of other patients |
4.4 |
4.3 |
4.2 |
| Getting information from others |
4.4 |
4.4 |
4.4 |
Table 3. Mean program materials enjoyment ratings of participants
(1 = did not enjoy; 5 = enjoyed very much) |
| Material |
Used material n (%) |
Mean Likert score for users |
| |
Men
(n = 65) |
Women
(n = 102) |
Men
(n = 65) |
Women
(n = 102) |
| Participating doctor |
65 (100%) |
102 (100%) |
4.2 |
4.2 |
| Program workbook |
28 (43%) |
44 (43%) |
3.3 |
3.9 |
| Ongoing tips |
37 (57%) |
38 (37%) |
3.8 |
3.9 |
| Fridge magnets |
40 (62%) |
40 (39%) |
3.4 |
3.5 |
| Website |
25 (38%) |
30 (29%) |
2.4 |
3.4 |
| Self-tests |
21 (32%) |
29 (28%) |
3.4 |
3.3 |
Providers
Likert scale averages for evaluation of the program (1 = poor; 5 = great) are shown for 19 providers (12 AHP facilitators and seven GPs) in Table 4.
| Table 4. Mean Likert scale scores for provider satisfaction with the program (1 = poor; 5 = great) |
| |
Facilitators
(n = 12) |
General practitioners
(n = 7) |
| How do you rate the notion of pSMAs for weight control? |
4.5 |
4.3 |
| How do you rate the notion of pSMAs for other chronic diseases? |
4.9 |
4.6 |
| Rate your interest in running other programs such as this for chronic disease. |
5.0 |
5.0 |
| Rate your interest in getting training and accreditation as a lifestyle medicine practitioner for presenting different types of chronic disease in a pSMA program like this, where the individual programs are written (in a standard format) by experts in each area. |
5.0 |
5.0 |
| pSMA, programmed shared medical appointments |
Common statements from providers about what they liked most about running pSMAs in weight control included:
Good change to the normal day – helps build more rapport with patients.
Seeing patients learning off each other is great.
It’s good to have time to cover topics and not repeat oneself.
Common statements about what they liked least included:
Financial concerns [for doctors]. (For example, is payment enough for the groups?)
No-shows and time required chasing people up.
Too many and too high level resources for some. These need to be simplified.
Body weight changes
Overall, participants lost an average of 3.2% (95% confidence interval [CI]: 2.2, 4.3; P <0.001) of their baseline weight at 12 months (Table 5). Male adherers lost 4.3% (95% CI: 2.6, 6.1; P <0.001) and female adherers lost 4.2% (95% CI: 2.7, 5.7; P <0.001) of their baseline weight at 12 months (Table 5; Figures 1 and 2). There were no significant changes among non-adherers.
Results obtained using BOCF were similar to analyses of the observed data, suggesting the results were not substantively affected by loss to follow-up.
Weight losses of >5% maintained from baseline are considered clinically significant.17 In the current study, 46% (95% CI: 33.1, 58.5) of male and 35% (95% CI: 25.5, 44.8) of female adherers achieved this (Table 6).
| Table 5. Percentage change in mean weight (kg) from baseline to 12 months with 95% confidence intervals by all participants, sex and adherer status |
| |
Baseline |
12 months observed data |
12 months baseline observation carried forward† |
|
Group
|
n (%) |
Mean weight (kg) |
n (%) |
Mean weight (kg) |
% change
from baseline
(95% CI) |
P value |
n (%) |
Mean weight (kg) |
% change
from baseline
(95% CI) |
P value |
| All participants |
216 (100%) |
105.4 |
192 (89%) |
102.2 |
–3.2 (–4.3, –2.2)* |
<0.001 |
216 (100%) |
101.7 |
–3.5 (–4.3, –2.7) |
<0.001 |
| Sex |
| Men |
83 (100%) |
114.1 |
78 (94%) |
110.6 |
–3.1 (–4.6, –1.5) |
<0.001 |
83 (100%) |
110.1 |
–3.6 (–4.8, –2.3) |
<0.001 |
| Women |
133 (100%) |
99.9 |
114 (86%) |
96.5 |
–3.4 (–4.8, –2.0) |
<0.001 |
133 (100%) |
96.4 |
–3.4 (–4.4, –2.5) |
<0.001 |
| Completed ≥4 sessions |
| Adherers |
166 (100%) |
105.2 |
153 (92%) |
100.7 |
–4.3 (–5.4, –3.1)* |
<0.001 |
166 (100%) |
100.4 |
–4.5 (–5.4, –3.7) |
<0.001 |
| Non-adherers |
50 (100%) |
105.8 |
39 (78%) |
108.1 |
0.4 (–1.7, 2.6)* |
0.684 |
50 (100%) |
105.8 |
0.0 (–1.0, 1.0) |
0.974 |
| Sex and completion of ≥4 sessions |
| Male adherers |
64 (100%) |
113.5 |
59 (92%) |
108.5 |
–4.3 (–6.1, –2.6) |
<0.001 |
64 (100%) |
108.0 |
–4.8 (–6.3, –3.4) |
<0.001 |
| Female adherers |
102 (100%) |
100.1 |
94 (92%) |
95.9 |
–4.2 (–5.7, –2.7) |
<0.001 |
102 (100%) |
95.7 |
–4.3 (–5.4, –3.3) |
<0.001 |
|
Male non-adherers
|
19 (100%) |
116.4 |
19 (100%) |
117.0 |
0.6 (–0.7, 1.9) |
0.382 |
19 (100%) |
117.0 |
0.6 (–0.7, 1.9) |
0.382 |
|
Female non-adherers
|
31 (100%) |
99.3 |
20 (65%) |
99.6 |
0.3 (–3.6, 4.4) |
0.875 |
31 (100%) |
98.8 |
–0.4 (–1.9, 1.0) |
0.554 |
*Adjusted for sex
†For each participant with missing weight measurements, their baseline value was used to fill in missing values at 12 months.
P values are for test of whether percentage change in weight is significantly different
CI, confidence interval |
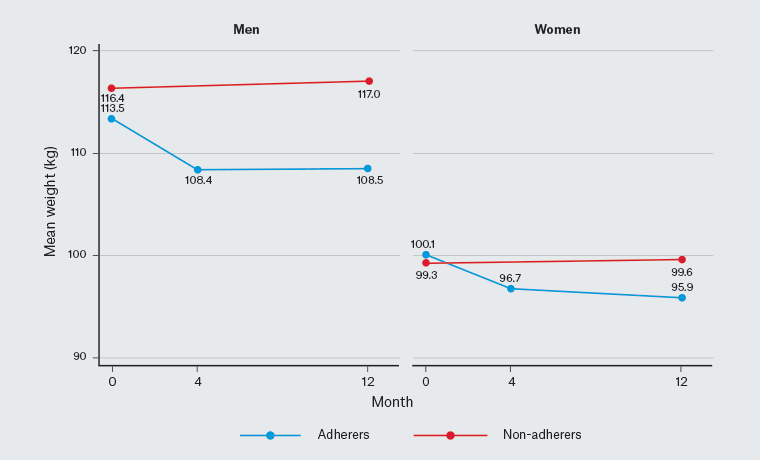
Figure 1. Mean weights (kg) over follow-up time by sex and adherer status
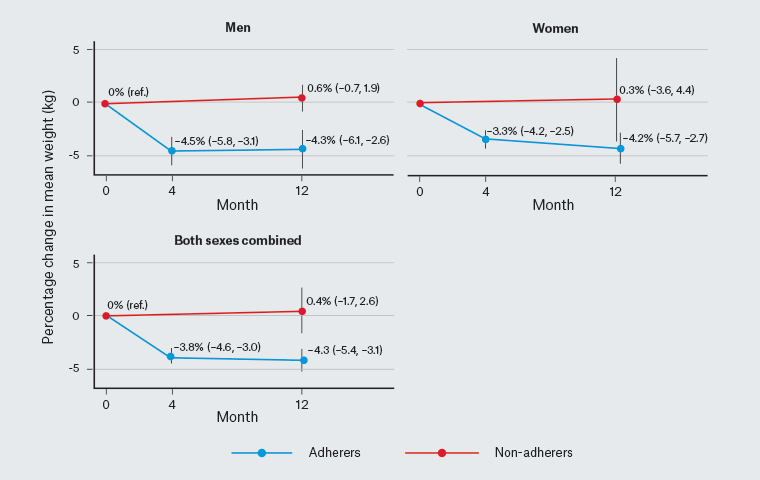
Figure 2. Percentage change in mean weight (kg) over follow-up time and 95% confidence intervals by sex and adherer status
| Table 6. Proportions of patients who achieved clinically important weight loss (>5%) from baseline to 12 months |
| |
Achieved clinical weight loss at 12 months |
| |
Observed data |
All participants* |
| Group |
Proportion |
% (95% CI) |
Proportion |
% (95% CI) |
| All participants |
63/192 |
32.8% (26.2, 39.5) |
63/216 |
29.2% (23.1, 35.2) |
| Sex |
| Men |
28/78 |
35.9% (25.3, 46.5) |
28/83 |
33.7% (23.6, 43.9) |
| Women |
35/114 |
30.7% (22.2, 39.2) |
35/133 |
26.3% (18.8, 33.8) |
| Completed ≥4 sessions |
| Adherers |
60/153 |
39.2% (31.5, 47.0) |
60/166 |
36.1% (28.8, 43.5) |
| Non-adherers |
3/39 |
7.7% (0.0, 16.1) |
3/50 |
6.0% (0.0, 12.6) |
| Sex and completion of ≥4 sessions |
| Male adherers |
27/59 |
45.8% (33.1, 58.5) |
27/64 |
42.2% (30.1, 54.3) |
| Female adherers |
33/94 |
35.1% (25.5, 44.8) |
33/102 |
32.4% (23.3, 41.4) |
| Male non-adherers |
1/19 |
5.3% (0.0, 15.3) |
1/19 |
5.3% (0.0, 15.3) |
| Female non-adherers |
2/20 |
10.0% (0.0, 23.1) |
2/31 |
6.5% (0.0, 15.1) |
*Patients who had missing weight values at 12 months were treated as having not achieved clinical weight loss.
CI, confidence interval |
Cost analysis
Using pSMAs for six sessions (10 participants/session), and a mean loss of 4.5 kg among adherers as reported here, with doctors’ time 1.5 hours/session, and assuming payment for the doctor of approximately $40 per person [pp] (current Medicare Benefits Schedule [MBS] item 23), and nurse $10 pp (ie a total of approximately $50 pp/session), the costs would be $300 pp or approximately $67/kg/pp. The time required for the doctor would be 14 mins/kg/pp.
Discussion
To the best of our knowledge, the current study is the first to report results of pSMAs for chronic diseases and conditions in primary healthcare; in this case, the factor examined was weight control. Our findings of positive participant and provider satisfaction, clinically significant weight loss outcomes in approximately 40% of adherers over 12 months and a significantly reduced cost and time effectiveness all support the process. An adherence rate of 77%, together with willingness to recommend the program to others and high ratings on aspects of the program, add to this conclusion.
There are three commonly quoted participant advantages of SMAs:2
- peer support and contribution of peers to the consultation
- extra time with a more relaxed and focused physician
- the contribution of AHPs to a consultation.
Almost three in four of those involved in this study said they ‘maybe/would’ prefer this type of consultation (for weight control) over a conventional 1:1 session with their doctor.
All 19 providers (facilitators and doctors) involved in the study also expressed satisfaction with the process, because:
- it reduces repetition to patients
- there is less pressure on the doctor
- the doctor is not focused on a computer screen (patient notes are recorded by the facilitator).
All providers agreed they would ‘definitely’ be interested in running pSMAs for other chronic diseases.
Overall mean percentage weight loss at 12 months was modest at around 3.2% (although approximately 4.3% among adherers). However, as clinically significant weight loss is regarded to be >5% of baseline weight,16 the proportions of adherers achieving this (46% of men and 35% of women) are of interest. Even with the most conservative analysis combining adherers and non-adherers, and assuming those lost to follow-up did not lose weight, 34% of men and 26% of women achieved clinically significant weight loss.
One US review has reported that a total of 11–26 (mean = 18) intensive visits with a doctor on a 1:1 basis over one year results in an average 4–7 kg (mean = 5.5 kg) weight loss.17 At 30 minutes per visit (nine hours total), using reimbursement rates in 2019 (ie approximately $75 per 30 mins), this would cost approximately $1350 pp, or $245/kg/pp, and take 98 mins/kg/pp. Comparative costs shown here are $300 pp or approximately $67/kg/pp and 14 mins/kg/pp. This is approximately four times more cost effective (approximately 27% of the cost of the conventional approach pp) and seven times more time effective (completed in approximately 14% of the time pp). Similarly, when compared with meta-analyses of commercial weight loss programs, the outcomes shown here over one year are up to 10 times more cost effective,18 and retention levels are greater (77%, compared with <70%).19 It is acknowledged, however, that 1:1 and SMA visits may be totally different when comparing the quality of care. Therefore, further work will be necessary to tease out these cost comparisons. These findings must also be considered preliminary to reflect any dynamic changes presenting to practice following the SMA process. More specific economic research is required here.
The current study was funded by a Primary Health Network (PHN) grant and did not rely on the MBS. The case for pSMAs may become more compelling in a capitation-funded system such as New Zealand, the National Health Service (NHS) in the UK, or the Health Care Homes model currently being trialled in Australia, where bundled payments provide greater certainty of funding as well as greater scope for multidisciplinary involvement. Cost effectiveness may also come from a lower burden of disease and decreased impact of healthcare costs in the future, which can only be determined by a long-term economic study. The SMA process covers the three main components of quality in healthcare (‘effective’, ‘safe’ and ‘client-centred’) as identified in a recent New England Journal of Medicine Catalyst survey.20
The primary limitation of this exploratory study was the single-arm design involving patients who may be more motivated to lose weight than typical overweight and obese patients. Similarly, while adherers lost considerably more weight than non-adherers, these differences in weight loss may be at least partly due to adherers being inherently more motivated than non-adherers. Despite these limitations, the degree of weight loss observed in this exploratory study is encouraging and warrants further investigation in a clinical trial randomising patients to SMA or conventional 1:1 clinical care. When designing such a trial, it is worth noting that post-hoc evaluations with providers and some participants suggest that outcomes might be improved further by changes to the program such as: fortnightly, rather than monthly, sessions; more specific up-skilling of doctors to ensure greater knowledge in evidence-based aspects of weight control; and better targeting of participants.
Chronic diseases in primary healthcare are unlikely to be managed fully through a single treatment modality. A procedure such as that tested here (pSMAs) could help a significant proportion (perhaps the majority) of patients with chronic diseases or ailments at the non-critical end of an affected population. If achieved at scale, pSMAs could enhance the contribution of PHC centres to population health outcomes.
Implications for general practice
- pSMAs are a series of SMAs that provide an alternative to conventional 1:1 clinical care.
- This exploratory study of pSMAs in weight loss shows they are highly popular among both participants and providers, with significant clinical outcomes after 12 months and cost and time efficiencies over conventional care.
- pSMAs provide an effective adjunct approach to chronic disease management in primary care.
SENSW Weight Management Trial Group
Includes the following SENSW Australia medical practices: Bombala St., Surgery, Cooma; Cringila Medical Centre; Milton Medical Centre; North Nowra Medical Centre; OCHRE Medical Centre, Wollongong; Russel Vale Family Medical and Acupuncture Practice, Wollongong; Sapphire Medical Clinic, Merimbula